Chiral LC-MS Analysis of Methamphetamine in Urine on Astec® CHIROBIOTIC® V2
Differentiation of Illicit D-Methamphetamine from Over-the-Counter L-Methamphetamine by LC-MS
Product No. 15024AST
Methamphetamine (Figure 1) is a powerful stimulant, often used as a recreational drug of abuse or as a doping agent in sports. Its presence in urine is screened for by enzymatic immunoassays and confirmed using a hyphenated mass spectrometry chromatographic method. However, this molecule exists in two enantiomeric forms, L-methamphetamine (or levo-methamphetamine), a vasoconstrictor used in the formulation of over-the-counter medications such as Vicks® Vapor Inhaler and D-Methamphetamine, the illicit stimulant.1 Immunoassay does not differentiate between the legal and illicit versions and therefore will report a positive finding if either are detected in the specimen above cutoff concentrations. The same holds true for reverse-phase LC-MS techniques that are commonly used in toxicology for drug of abuse quantitation.
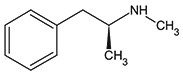
D-Methamphetamine
(dextro-Methamphetamine,
S(+)-Methamphetamine)
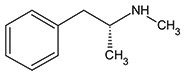
L-Methamphetamine
(levo-Methamphetamine,
R(-)-Methamphetamine)
Figure 1. Structure of D- and L- Methamphetamine
This poses a problem when analyzing specimens from patients who are using the legal decongestants or medications such as selegiline and fenproporex, which may metabolize to methamphetamine.2 To confirm which molecule is present in a specimen testing positive by immunoassay for methamphetamine, a chiral LC-MS method was developed using an Astec® CHIROBIOTIC® V2 HPLC column. The method was applied to real patient samples allowing such differentiation with an extremely high level of accuracy.
Experimental
Certified Reference Material standards (rac)-methamphetamine and (L)-methamphetamine were purchased from Cerilliant, TX, U.S.A. Urine samples were obtained from patients visiting pain management clinics and as part of a random drug-testing program aimed at monitoring for prescription compliance. All specimens were stored at -4 °C and thawed on the morning prior to analysis. For analysis by chiral LC-MS/MS, samples were processed as follows: To a 125 µL aliquot was added 1 mL of diethyl ether. The diluted sample was vortexed for 30 minutes and centrifuged at 10,000 rpm for 10 minutes. A 500 µL aliquot of the organic supernatant was evaporated to dryness at 56 °C in a heating block. The dried extracts were resuspended in 0.5 mL of starting mobile phase and 10 µL were injected onto the column. Analysis of patient samples was preceded by the injection of an authentic spiked standard sample containing both enantiomers in methanol (D:L, 1:3).
The liquid chromatography system (Waters® ACQUITY® UPLC®, Waters, Milford MA, U.S.A.) was run in isocratic mode, without temperature control using an Astec® CHIROBIOTIC® V2 column (25 cm x 4.6 mm, 5 µm) from Merck The mobile phase consisted of 0.04% w/v ammonium trifluoroacetic acid in water:methanol (5:95, v/v) and the flow rate was set at 1 mL/min. Mobile phase is prepared by dissolving 0.4 g ammonium trifluoroacetate into a solution of 950 mL methanol and 50 mL water and mixing well. Total run time was 13.00 minutes. Retention times were 10.75 and 11.62 min for D- and L-methamphetamine, respectively. The mass spectrometer (MS, Waters) was operated in ESI+ and MRM modes. The two following transitions were monitored for positive identification of both enantiomeric compounds, 150.0..91.0 and 150.0..119.0. Peak smoothing and integration were carried out to determine relative ratios using the MassLynx™ software. D- and L- peak assignments were established by comparison of retention times with the spiked standard sample.
Results
Chiral separation of D- and L-methamphetamine using the Astec® CHIROBIOTIC® V2 column and the methanol-ammonium trifluoroacetic acid mobile phase was successfully applied to the analysis of real patient urine samples that had previously been identified as positive for methamphetamine by quantitative analysis on a C18 column. Figure 2 provides chromatographic traces of standards as well as several patient samples. Trace 1 in the figure shows a 1:3 standard mixture of D- and L-methamphetamine, respectively. Traces 2 through 4 show the response of D-methamphetamine in patient samples, indicating illicit use of the drug by these patients. The L-methamphetamine response in Trace 5 indicates legal use of OTC-derived L-methamphetamine.
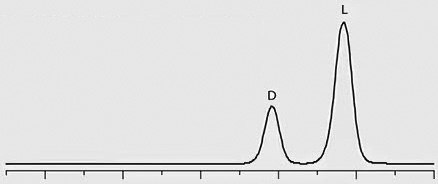
Trace 1: 1:3, D:L-Methamphetamine Standard
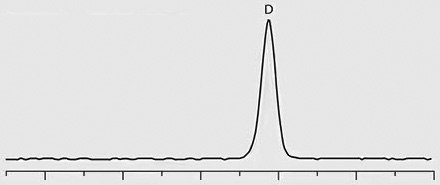
Trace 4: Patient Sample – ILLICIT, D-Methamphetamine Positive
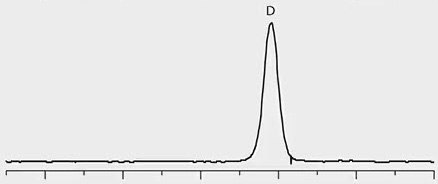
Trace 2: Patient Sample – ILLICIT, D-Methamphetamine Positive
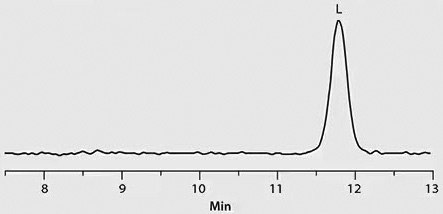
Trace 5: Patient Sample – Legal, L-Methamphetamine Positive
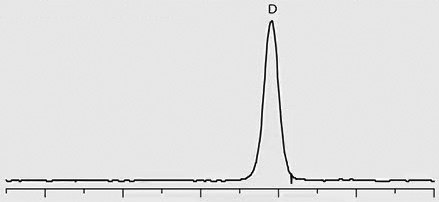
Trace 3: Patient Sample – ILLICIT, D-Methamphetamine Positive
Figure 2. Chiral LC-MS/MS Separation of D- and L-Methamphetamine Enantiomers on Astec® CHIROBIOTIC® V2.
Conclusion
Screening studies for illicit drugs require confirmation. In the case of methamphetamine, the positive results from screening studies are often the result of the metabolism of prescribed therapeutics or the inability of the method to differentiate between D-methamphetamine and the enantiomer, L-methamphetamine, found in OTC medications. Astec® CHIROBIOTIC® V2 is shown to efficiently separate methamphetamine enantiomers using LC/MS-compatible conditions allowing sensitive differentiation of stereoisomer forms present in urine.
References
如要继续阅读,请登录或创建帐户。
暂无帐户?