Tips and Techniques for Troubleshooting Immunohistochemistry (IHC)
Immunohistochemistry (IHC) is a technique in which antibodies are used to detect an antigen in sectioned tissue and is ubiquitous in the research, preclinical, and clinical settings for visualizing cellular components. This technique, which was first used in the 1940s, is widely used to increase understanding of the distribution and localization of differentially expressed proteins in tissues.
The steps for executing IHC and many of the tools and reagents have remained relatively unchanged for decades. However, understanding the basic steps in a simple IHC protocol is no guarantee of consistent, interpretable images. Optimization and fine-tuning are the keys to success in developing an IHC protocol for a new target.
The generalized steps of an IHC protocol are simple:
- Specimen Preparation
- Antigen Retrieval
- Blocking
- Primary Antibody Staining
- Detection
The steps seem simple, but optimization of your protocols and reagents can be the difference between no staining at all and bold staining that can alter the path of your research. Below, our R&D histologists and antibody validation scientists offer some insights into the optimizations that will help you avoid common IHC problems in order to get the most useful data and publishable images, regardless of your choice of technique.
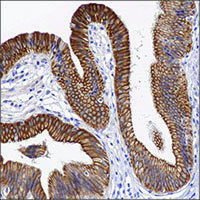
Figure 1.Light microscopy image of anti-PIR antibody validation by IHC on FFPE human bladder sections using diaminobenzidine (DAB) enzymatic detection (brown chromogen) and counterstaining with hematoxylin (blue).
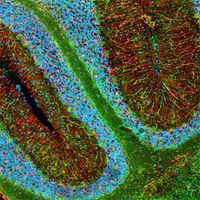
Figure 2.Fluorescent microscopy detection of non-phosphorylated rat neurofilament H (Cat. No. NE1023, green) by fluorescent staining of frozen sections of rat brain. Blue, Hoechst 33342.
Specimen Preparation: The Foundation of Successful IHC
Regardless of preservation method or microscopic analysis approach, most histologists would agree that appropriate sample preparation, which preserves not only tissue morphology but also the target epitope(s), is the foundation for any successful IHC procedure. The protocol varies significantly depending on whether analysis by fluorescence or light microscopy is planned.
Tissue Collection and Fixation
Appropriate and efficient tissue collection and preservation for IHC safeguards cell and tissue morphology while halting proteolytic and microbial degradation. The two principal methods for tissue preservation for IHC are rapid freezing and FFPE (formalin fixed, paraffin embedded).
In frozen tissue preparations, tissue is rapidly harvested into a nonaqueous medium, and subsequently sectioned at very cold temperatures on a cryostat. FFPE preparations offer the convenience of indefinite storage and sectioning of tissue embedded in paraffin blocks at room temperature on a microtome.
Cryopreservation
Immediately following dissection, fresh tissue should be quickly frozen in chilled isopentane (2-methylbutane) or an equivalent alternative. Rapid freezing is essential to prevent the formation of ice crystals that can damage tissue. Frozen tissue is then stored at -80 °C until sectioned. For large tissues such as human organs, tissue must first be sectioned into smaller blocks to facilitate faster freezing that is uniform throughout the tissue volume.
After thawing and prior to cutting, frozen tissue is embedded in molds with a freezing medium which is also used to adhere it to the cryostat chuck and allow the frozen tissue to contact the sectioning blade in a controlled manner. Tissue should also be allowed to equilibrate in the cryostat chamber for 30 minutes prior to cutting to facilitate the acquisition of consistent and representative tissue slices. The optimal cryostat temperature for sectioning depends on the tissue type and freezing protocol. Be mindful to keep track of tissue orientation during all stages of cryostat sectioning. After each section is made, gently lay the section out on a frozen glass slide. The use of a frozen paintbrush and an anti-roll tool (along with lots of practice) will help avoid tissue section rolling, folding, wrinkling, and tearing.
Antigen Retrieval
Although FFPE is an effective and reliable method for ensuring the preservation of tissue morphology for extended periods at ambient temperatures, this format simultaneously renders many target epitopes inaccessible to antibody detection. Antigens in FFPE tissue sections may be unmasked by one of these retrieval methods:
- HIER (heat-induced epitope retrieval): Tissue sections on slides are subjected to a specific sequence of heating with simultaneous pressure increase in a citrate buffer. This is achieved in a device similar to a pressure cooker
- PIER (protease-induced epitope retrieval---also called EIER, enzyme-induced epitope retrieval) refers to the employment of an enzyme—proteinase K, trypsin, or pepsin—incubated at 37 °C. TIP: For a few slides, the enzyme solution can be added directly to slides; in most cases, however, reagent concentration, temperature and incubation time are best controlled by simultaneous immersion of all slides to be processed in a bath containing the enzyme.
Staining
Selecting Antibodies for IHC
Specificity: Primary antibody selection is a critical step for effective IHC. One especially important consideration is antibody specificity, which ensures that the antibody binds only to the protein of interest. A gold standard for assessing specificity is the finding that staining is absent in tissue from which the protein of interest has been knocked out, although other methods, such as the presence of a single band using Western blotting, also exist. Selection of a secondary antibody should always ensure that the secondary is specific to the species that the primary antibody was originally obtained from. Several additional factors can each determine the efficacy of the secondary antibody and include, epitope specificity to the primary antibody, purification methods, and if the antibody is absorbed against cross-reactive immunoglobulins.
Application suitability: Reputable antibody suppliers will typically indicate immunodetection applications for which an antibody has been validated, and should supply data to demonstrate results for IHC/ICC (immunocytochemistry). For example, antibodies that successfully label an epitope under the denatured conditions of Western blotting may not necessarily perform during IHC, in which the native protein conformation is often preserved.
Clonality: Monoclonal antibodies are derived from expanding a single plasma cell, and therefore recognize a unique epitope. While monoclonal antibodies offer specificity, polyclonal antibodies can be a better choice when the protein target is expressed at low levels, as they recognize multiple epitopes and therefore offer greater sensitivity. Conversely, the manufacturing process for polyclonal antibodies makes them intrinsically subject to greater lot-to-lot variation within product specifications.
Host source: A fourth important antibody consideration is the host species that was used to generate the antibody. To avoid cross-reactivity, it is essential that the antibody be raised in a different tissue than that of the tissue under study. The primary antibody host species will also need to be matched to the secondary antibody during detection.
Blocking: Non-specific binding of the antibody is of paramount concern when it comes to immunodetection of the antigen by the primary antibody. However, non-specific detection of antigens by the secondary can also lead to significant background after the substrate addition step. A critical step to reduce background staining and ensure antigen specificity is by incorporating a blocking step prior to the addition of the primary antibody. The blocking solution should ideally contain serum that matches the species of the secondary antibody.
Detection
Detection is typically achieved using one of two methods: (a) colorimetric or enzyme-mediated detection and (b) fluorescence-based detection.
In the colorimetric method, the bound primary or secondary antibody is conjugated to a substrate which yields a precipitating product when converted by an enzyme. This precipitate is visible as colored staining when viewed by light microscopy.
In the fluorescence-based detection method, antibody bound to the antigen of interest in the tissue is directly or indirectly conjugated to a fluorophore (also sometimes called a fluorochrome), a molecule that fluoresces in the presence of light of a specific wavelength.
Analysis of Results: Fluorescent vs. Light Microscopy
Fluorescent Microscopy
Both immunofluorescent ICC and IHC require an understanding of the configuration of epifluorescence or confocal microscope that will be used to analyze the sample. The best results will be obtained when spectral characteristics of the fluorophores conjugated to the primary or secondary antibody are matched to the excitation source (usually a laser) and emission filters of the available microscope. For more about designing a fluorescence microscopy analysis experiment, visit this page.
Light Microscopy
White light or brightfield microscopy may be more accessible to researchers as the necessary equipment is available in most labs, but is limited by the number of targets which may be simultaneously detected in the same tissue section. This is because deposition of the chromogen at the site of antibody binding to antigen in the tissue is dependent on enzyme-substrate activity, and the most common, reliable reagents (such as diaminobenzidine, or DAB, and alkaline phosphatase, or AP) do not offer multispectral colorimetric detection that would permit simultaneous detection of multiple targets.
Troubleshooting Problems with IHC Results
The following table presents some common problems encountered with IHC results, along with possible explanations and suggestions for technical correction/optimization.
Products
如要继续阅读,请登录或创建帐户。
暂无帐户?