Membrane Integrity Test for Lipid-PAMPA Artificial Membranes
Background
The Parallel Artificial Membrane Permeation Assays (PAMPA) is a non-cell based assay designed to predict passive, transcellular permeability of drugs in early drug discovery. The assay is carried out in a 96-well MultiScreen® PAMPA filter plate (MAIPN4550 or MAIPNTR10) and measures the ability of compounds to diffuse from a Donor compartment to an Acceptor compartment separated by a PVDF membrane filter pretreated with a lipid-containing organic solvent.
One distinct advantage of the lipid-PAMPA assay over similar permeability assays such as HDM-PAMPA is the ability to tailor the artificial membrane lipid components and relative concentrations to mimic the composition of specific target barriers such as the intestinal lining or blood brain barrier. While a customizable artificial membrane is a highly desirable feature, there remains the potential for data unreliability due to difficulties in formulating and applying the lipid/organic solvent mixtures to the PVDF membrane filters. Similar permeability assays such as HDM-PAMPA and Caco-2 have simple, well established procedures which are capable of eliminating spurious data caused by non-integral artificial membranes or cell layers. TEER testing (Trans Epithelial Electrical Resistance), which measures the electrical resistance across a membrane, is a rapid and straight forward method for measuring membrane integrity of both HDM-PAMPA membranes and cell monolayers. Additionally, Lucifer Yellow rejection is a common analytical method for determining Caco-2 cell layer integrity; however, neither membrane integrity test is completely applicable to Lipid-PAMPA membranes. This protocol note outlines a method by which the integrity of the lipid membrane can be assessed immediately following drug permeation assays. Upon completion of the lipid-PAMPA assay, lipid membrane integrity is determined based on the transport of two compounds, Brilliant cresyl blue which has a low permeability and Lucifer Yellow which has very poor permeability and should effectively be completely rejected. A lipid membrane that is uniform and integral will result in the passage of some Brilliant cresyl blue to the acceptor and complete rejection of Lucifer Yellow. The final acceptor solution should have a visible blue tint and fluorescence values equal to background standards. Failure modes (e.g. PVDF was not completely wet out, air locking, a physical defect in the PVDF membrane) will result in either passage of Lucifer Yellow and/or rejection of Brilliant cresyl blue in high quantities or complete rejection of both compounds.
Experimental Materials
Reagents
- Phosphate buffered saline (# P-3813; Sigma)
- Dimethyl sulfoxide (# D136-1; Sigma)
- Brilliant cresyl blue (# B-2002; Sigma)
- Lucifer yellow (# L0144; Sigma)
Equipment
- MultiScreen PAMPA assay plate (# MAIPN4550 or MAIPNTR10; Millipore Corporation, Billerica, MA)
- PTFE Acceptor plate (# MSSACCEPT0R; Millipore)
- Spectramax® Gemini fluorescence microtiter plate reader (Molecular Devices, Sunnyvale, CA) or similar instrumentation
- Spectramax® Plus microtiter plate reader (Molecular Devices, Sunnyvale, CA) or similar instrumentation
- SoftMax® Pro software (Molecular Devices)
- Multichannel pipette such as:
- Finnpipette® electronic pipettor (# 21377232; Thermo lab Systems, Helsinki, Finland)
- Biohit™ 8 channel electronic pipettor with polypropylene tips (# W67-710-800, W16-160045, Vanguard International, Neptune, NJ)
- Stericup® with 0.22 µm, GP Express Plus (# SCGPU10RE; Millipore Corporation, Billerica, MA)
Protocol
- Prepare 1 L of a 5% solution (v/v) of DMSO in phosphate buffered saline pH 7.4.
- Add 0.1 g of Brilliant cresyl blue to 1 L of 5% DMSO in PBS and mix the solution for a minimum of 1 hour.
- Filter the Brilliant cresyl blue stock solution through a Stericup with 0.22 µm GP Express Plus membrane to remove any remaining solid Brilliant cresyl blue.
- Add 100 mg of Lucifer yellow to 100 mL of the Brilliant cresyl blue (BCB) stock solution and mix until completely dissolved to create the Lucifer yellow (LY) stock solution.
- Prepare the final Brilliant cresyl blue/Lucifer yellow test solution by mixing the two stock solutions in a 9:1 ratio, 9 parts BCB to 1 part LY (~15 mL/plate required).
- Upon completion of the PAMPA assay rinse out the acceptor plate and remove any remaining liquid in the donor wells by gently inverting the plate and shaking (Note 1).
- Add 300 µL of buffer (5% DMSO in PBS pH 7.4) to each well of the PTFE acceptor plate.
- Add 150 µL of the BCB/LY test solution to each well of the donor plate and assemble the donor/acceptor plate “sandwich”.
- Replace the plate lid and incubate for 3 to 4 hours.
- After incubation, analyze the acceptor solution to determine the presence of Brilliant cresyl blue. The solution will have a blue tint if BCB successfully permeated across the lipid membrane (Note 2).
- Using a fluorescence microtiter plate reader (excitation 425 nm – emission 528 nm), measure the fluorescence of the acceptor solution to verify Lucifer Yellow rejection by the membrane (Note 3).
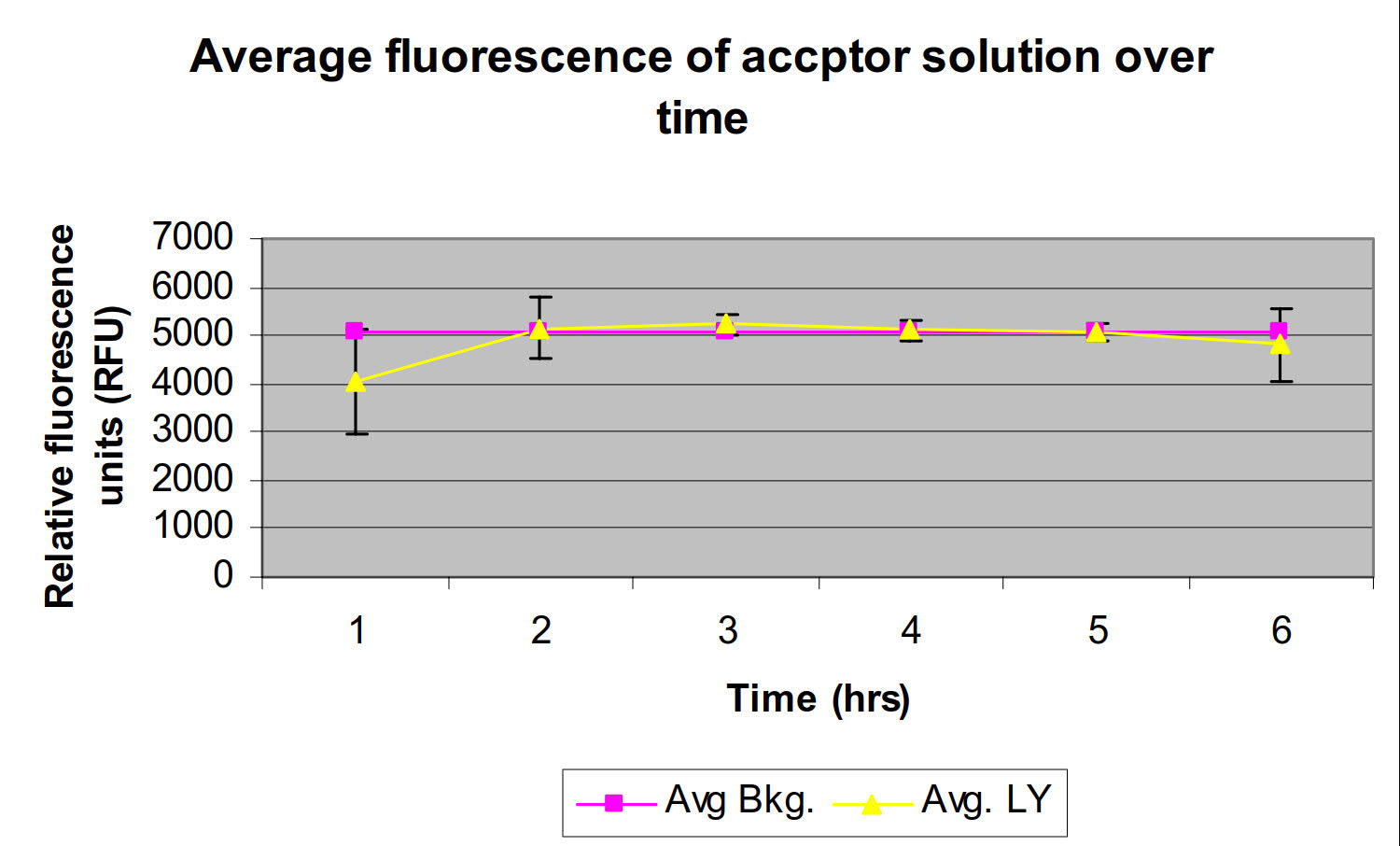
Figure 1.Lucifer yellow rejection by 2% DOPC in dodecane over time.
Materials
如要继续阅读,请登录或创建帐户。
暂无帐户?