LC-UV-MS Method Development for Antibody Drug Conjugates Using a Non-Toxic ADC-Mimic
Introduction
Antibody-drug conjugates (ADC) represent a rapidly emerging class of biotherapeutic molecules, which harness the specificity of monoclonal antibodies (mAb) to selectively deliver highly potent cytotoxic drugs. Like all biotherapeutics they require significant analytical characterization. A non-toxic ADC surrogate, or “ADC Mimic,” with highly similar physicochemical properties to commercial ADCs provides an excellent molecule to safely drive analytical method development for ADCs.
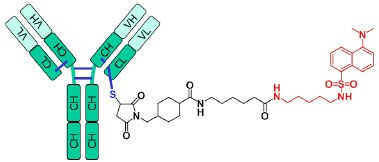
Figure 1.ADC Mimic
ADC Mimic – MSQC4 plus 2, 4, 6, or 8 dansylcadaverine- SMCC mimic-linkers. Each conjugation to a Cys residue imparts a +668 Da mass shift.
Here, we build on previous work describing the preparation of such a non-toxic ADC-Mimic by utilizing the ADC-Mimic and the underlying mAb (MSQC4) to perform a comparison of chromatographic stationary phases. We also present the use of the ADC-Mimic to develop a workflow to characterize ADC variants by first separating them via hydrophobic interaction chromatography (HIC), collecting fractions, and subsequently analyzing the ADC variants by RP-LC-UV-MS.
Methods
MSCQ4 was reduced prior to analysis while the ADC Mimic was deglycosylated overnight with PNGase F and then reduced. Reduction was performed with 7.4 M GndCl and 100 mM DTT at 50 ºC. An Acquity-QToF Premier was used for the column comparison and mass confirmation. A Prominence (Shimadzu) was used for the HIC separation and fractionation. An M-Class Acquity-Xevo G2 QToF (Waters) was used to analyze the HIC fractions after deglycosylation and reduction.
- Bioshell A400 Protein C4 150 × 2.1 mm, 3.4 µm (Supelco)
A: 0.1% TFA in H2O; B: 0.1% TFA in ACN - PLRP-S 1000A 150 ×2.1 mm, 8 µm (Agilent)
A: 0.1% TFA in H2O; B: 0.1% TFA in ACN - TSKgel Butyl-NPR 100 x 4.6 mm (Tosoh)
A: 50 mM PO4 pH 7.0 , 1.5 M (NH4)2SO4 , 5% IPA
B: 50 mM PO4 pH 7.0 , 20% IPA
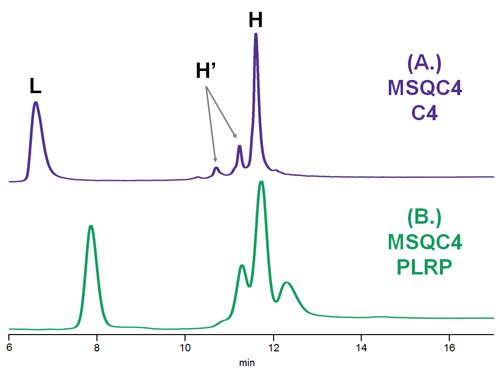
Figure 2.Comparison of C4 and PLRP columns using standard reduced mAb, where L = light chain; H = heavy chain; H’ = oxidized heavy chain.
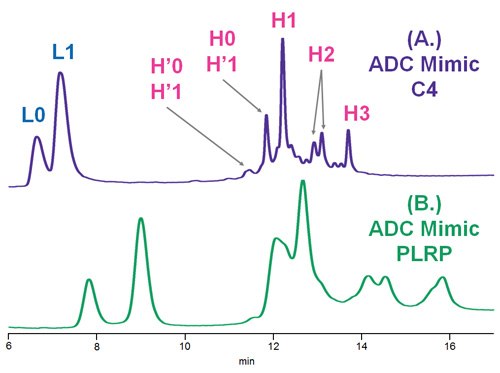
Figure 3.Comparison of C4 and PLRP columns using deglycosylated, reduced ADC Mimic, where L = light chain; H = heavy chain; H’ = oxidized heavy chain and the number represents the number of conjugations observed. MS confirmations of light and heavy chain species are shown in Figures 3 and 4.
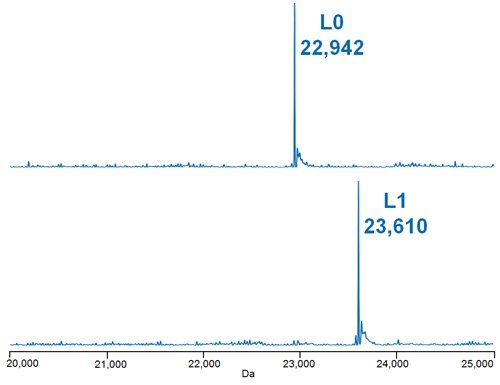
Figure 4.Deconvoluted mass spectra of the light chain species of the deglycosylated, reduced ADC Mimic shown in Figure 2 (A.), where L = light chain and the number represents the number of conjugations observed.
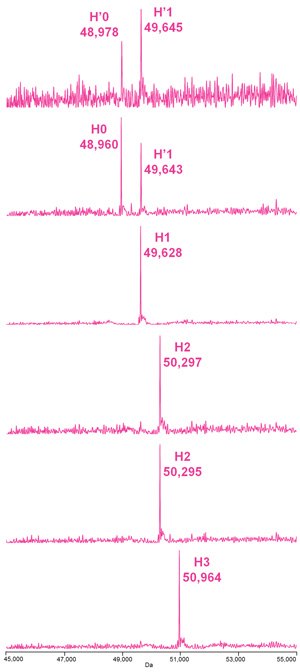
Figure 5.Deconvoluted mass spectra of the heavy chain species of the deglycosylated, reduced ADC Mimic shown in Figure 2 (A.), where H = heavy chain; H’ = oxidized heavy chain and the number represents the number of conjugations observed.
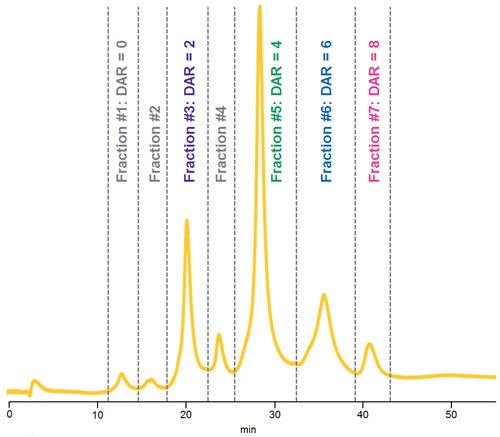
Figure 6.HIC separation of native ADC Mimic demonstrating fractions collected for LC-UV-MS analysis.
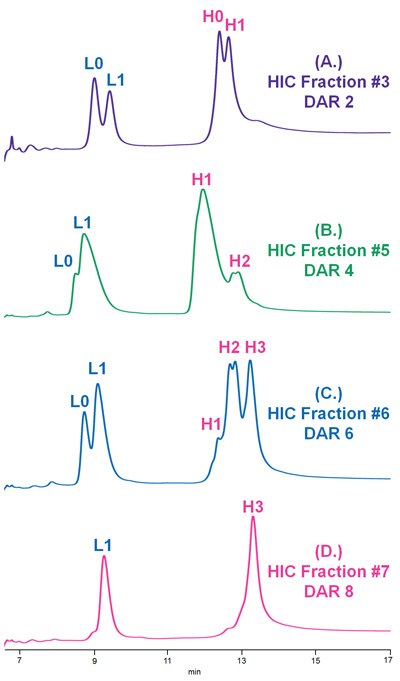
Figure 7.Reversed phase separation of collected HIC fractions pertaining to (A.) DAR 2, (B.) DAR 4, (C.) DAR 6, and (D.) DAR 8 ADC Mimic species, which were deglycosylated and reduced prior to LC-UV-MS analysis.
Summary
A non-toxic ADC Mimic was used to safely test chromatography conditions for developing workflows to characterize ADCs.
Mass spectrometry was used to confirm light chain and heavy chain species with zero or more conjugations of dansyl-cadaverine-SMCC mimic-linkers.
HIC fractionation and subsequent reversed phase LC-UV-MS allowed for structural confirmation of unique ADC Mimic species with different drug loads.
Materials
To continue reading please sign in or create an account.
Don't Have An Account?