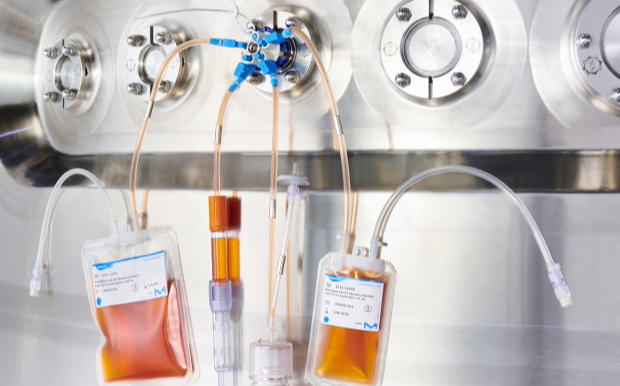
Sterile Process Sampling

A robust pharmaceutical or biopharmaceutical drug manufacturing process relies on reliable sampling from process development through commercial manufacturing. Closed, sterile sampling solutions enable accurate, representative sample collection and minimize the risks of process contamination.
NovaSeptum® sterile sampling solutions enable you to manually sample throughout your drug manufacturing to monitor both your product quality and process control. These sampling solutions are suitable for every process step including analysis of process analytes, measurements of product and process impurities, assessment of product quality attributes as well as sterility, bioburden, and endotoxin testing.
Key Benefits and Features
Quality you can trust: Leveraging our global quality systems and the Emprove® Program, our NovaSeptum® Sampling Systems come with extensive documentation and regulatory support, facilitating your risk assessment and process control.

Minimizes cross-contamination risk and protects the integrity of your samples and process.

Plug-and-play design with easy connectivity and multiple container options.

Multiple containers with different size options in single and manifold designs for maximum flexibility.
Sampling Solutions for Multi- and Single-Use Processes
NovaSeptum® sampling technologies offer closed, presterilized and easy to use options for both multi-use and single-use processes that ensure process integrity and representative sampling. With formats that include single units or manifolds, and range of containers to fit different sampling needs, these reliable sampling technologies can be preconfigured or configured on site depending on specific process needs.
NovaSeptum® GO Sampling Systems for multi-use processes are closed, easy to use systems that ensure process integrity and representative sampling with sampling actuation evidence and control.
NovaSeptum® SURe Sampling Assemblies offer a closed, representative sampling solution for single-use processes that can be easily integrated using AseptiQuik® connectors or heat weldable C-Flex® tubing.
Reliable Bioprocess Sampling
From bioreactor to final filling, NovaSeptum® sampling solutions provide options for consistent and representative sampling so you can assure product quality and demonstrate control of your manufacturing process. These sterile, manual sampling solutions can be used throughout process development and commercial manufacturing. They excel in environments where maintain sterility is paramount, making them ideal for sterility, bioburden, mycoplasma, and endotoxin testing.
Interested in automated sampling for increased sampling frequency and enhanced process insights? Discover the MAST® Autosampling Solution, ideal for applications requiring frequent sampling and close monitoring, especially those using process analytical technology (PAT). Need more information? Jump to our technical article Bioprocess Sampling Systems: Find the Right Sampling Approach for Your Needs
Frequently Asked Questions
What is closed sterile sampling and why is it essential in biopharmaceutical manufacturing?
Closed sterile sampling is the process of collecting samples from biopharmaceutical processes while ensuring the samples remain uncontaminated. This is crucial for maintaining product quality and compliance with regulatory standards, as it allows for accurate monitoring of critical quality attributes (CQAs) and critical process parameters (CPPs) process while maintaining sample source integrity. Learn more about in our sampling in pharmaceutical and biopharmaceutical processes dedicated technical article.
What are the key benefits of using NovaSeptum® sterile sampling solutions?
NovaSeptum® sterile sampling solutions provide reliable and representative sampling while minimizing cross-contamination risks. Their closed, easy-to-use designs allow for plug-and-play connectivity and supports multiple container options, ensuring maximum flexibility for both development and manufacturing environments. They are especially well suited whenever critical sterility, mycoplasma, bioburden, and endotoxin testing is required. Learn more about in our closed sampling and NovaSeptum® sampling solutions free on-demand webinar.
How does manual sterile sampling contribute to regulatory compliance?
Manual closed sterile sampling adheres to stringent guidelines set by regulatory bodies, ensuring that samples are collected and handled in a manner that minimizes contamination risks. Proper documentation and adherence to standard operating procedures (SOPs) during the sampling process are vital for demonstrating compliance during audits. Find out more about regulatory considerations related to sampling.
In what situations is manual sampling with NovaSeptum® sterile sampling solutions preferred over automated sampling?
Manual closed sterile sampling may be preferred in scenarios requiring flexibility, such as during development or when sampling from unique or non-standard setups. It is highly recommended for sterility, bioburden and endotoxin testing at any stage of drug development or in GMP manufacturing. Manual sampling can be beneficial in smaller-scale operations where automated systems may not be cost-effective or practical. Explore our technical article on risk mitigation through bioburden control.
Will manual sampling cover all my sampling needs?
Manual closed sterile sampling can be used for most sampling needs but for routine process monitoring where frequent data points are needed to improve process understanding, process control and maintain product quality, automated sampling systems and other process analytical technology (PAT) are a better option.
Related Webinars
In this webinar we will explore the regulatory framework that guides sampling methodologies and outline the key qualifications necessary for effective sampling assembly, including system integrity evaluations.
we will focus on an automated sampling system and its utilization in perfusion cell culture for upstream process development. We will demonstrate an increased frequency of data acquisition for the online measurement of CPPs and automation of a sample processing workflow to reduce the time to measure CQAs in perfusion cultures.
This webinar explores how automation strategies and control schemes facilitate rapid analytical results, crucial for managing glycosylation variability that affects product quality.
Related Articles
- Discover how Process Analytical Technology (PAT) enhances biopharmaceutical manufacturing through aseptic sampling and real-time quality assurance.
- Ensure sample integrity and accurate results with NovaSeptum® sterile sampling containers: bags, bottles, syringes & more.
- Symbolic depiction of a (bio-)pharmaceutical process with persons in lab coats working, possibly taking samples.
- Explore the importance of closed sampling in pharma manufacturing, focusing on regulatory guidance, bioburden control, and process monitoring.
- Explore microbial control strategies in drug manufacturing: bioburden management, contamination prevention, and reliable closed sampling.
- See All (6)
Related Documents
- White Paper: Aseptic Process Sampling Risk Mitigation – A Regulatory Perspective
There are significant consequences associated with microbial contamination during biopharmaceutical manufacturing. Contamination increases risks for the operator, the company, and potentially the patient, all of which can result in significant negative impact.
- Application Note: Microbial Integrity of NovaSeptum® Sampling Systems upon Performance of Multiple Sampling Actuations
The NovaSeptum® and NovaSeptum® GO sampling systems are a family of products designed for singleuse sterile sampling throughout the biomanufacturing process.
- User Guide: NovaSeptum® and NovaSeptum® GO Sampling Units
This user guide provides installation and use instructions
- Data Sheet: NovaSeptum® GO Sterile Sampling System
To demonstrate the safety and integrity of your product, you need a standard-setting sampling solution that provides the flexibility to sample throughout your entire process.
- Data Sheet: NovaSeptum® SURe Sterile Sampling Assemblies
NovaSeptum® SURe single-use sterile sampling assemblies are designed to protect your single-use process from potential cross-contamination ensuring representative samples.
- Brochure: NovaSeptum® GO Sterile Sampling System
Continuing to set the sampling standard, our NovaSeptum® GO sterile sampling system is equipped with additional features for even safer sampling throughout your entire process.