Improve Product Quality and Process Control with Sampling Solutions for Pharma and Biopharma

Manufacturing of any pharmaceutical or biopharmaceutical drug involves comprehensive sampling and testing throughout manufacturing to assure product quality, safety, and efficacy. For parenteral drug products that cannot undergo terminal sterilization, strict control measures to prevent introduction of contaminants throughout the process are especially critical.
Regulatory guidance documents outline testing expectations and emphasize the value of risk- and science-based approaches in control strategies to support aseptic drug manufacturing. Any control strategy must be aligned with risks associated with product quality and patient safety.1-5
Discover key aspects and considerations for sampling in a (bio-)pharmaceutical environment in this article.
Why is Pharma and Biopharma Process Sampling Performed?
In pharmaceutical and biopharmaceutical production, sample collection serves many purposes. Regardless of modality, unit operation, or stage of development, sampling assures alignment of product quality, safety, and efficacy to specifications and enables routine monitoring of critical process parameters (CPPs) and critical quality attributes (CQAs). Global shifts towards increased process understanding and control, along with prevalence of process analytical technologies (PAT) and integration of quality by design (QbD) principles into manufacturing has made sampling an essential tool in the control of drug manufacturing.
Robust sampling strategies are critical in assessing both the quality of the drug product and control of the drug manufacturing process.
Sampling technologies also offer opportunities for safely transferring material into processes while minimizing the risk of process contamination; for example, addition of supplements or other materials to cell culture processes or seeding by inoculation (Table 1). In addition, closed sampling technologies improve operator safety, which is especially important when highly toxic materials are involved, as is the case in antibody-drug conjugate (ADC) manufacturing.
When is Pharma and Biopharma Process Sampling Performed?
A robust sampling strategy should be scientifically sound, justifiable based on the process risks and detail of essential criteria such as sample collection method, the number and volume of samples taken, storage conditions, frequency of sample collection among others. This information provides confidence in the integrity of the sample and subsequent test results.
In aseptic drug manufacturing sampling is conducted on:
- Starting/raw materials: to confirm identity and quality, including bioburden
- Process intermediates: to confirm appropriate composition, levels of contaminants
- In-process monitoring: to monitor CPPs that would impact CQAs of the final product and process performance
- Final products: to confirm identity, purity, potency, safety and final release testing
Sampling strategies are highly dependent on the stage of development, analytical needs and individual process risks. Figure 1 provides an example of a simplified sampling strategy for a monoclonal antibody (mAb) mid- to large-scale process and illustrates critical lot release sampling and testing points.
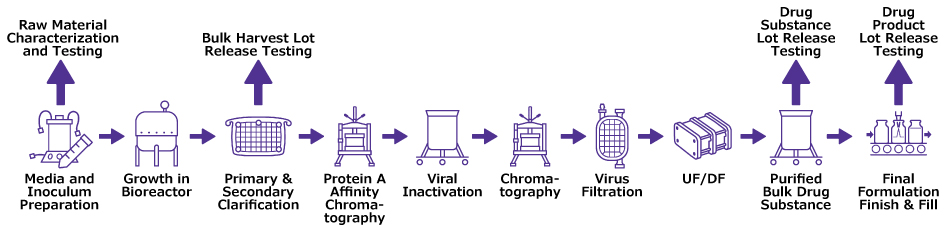
Figure 1.Simplified process flow and key sampling points for a typical mAb process
What Are the Requirements for Sampling Devices?
‘Traditional’ sampling methods used systems that were not closed, meaning they had no barrier to prevent microbial ingress to either the process or the sample during sample collection. Consequently, multiple microbial contaminations of sterile injectable drugs occurred resulting in drug manufacturers implementing corrective actions to avoid recurrence.6
The Shift to Closed Sampling Devices
The limitations of traditional sampling methods led regulatory bodies and industry groups to recommend closed aseptic sampling technologies. Closed sampling technologies mitigate the risk of microbial ingress, eliminate operator bias and errors and increase the likelihood that the sample is representative of the process. The WHO Guidelines list multiple requirements for aseptic sampling devices, Table 2.3
For additional in-depth information and expert insights on closed sampling, please access our on-demand webinar:
Closed Sampling: Crucial for Aseptic Processing in Manufacturing Quality and Regulatory Compliance.
Sampling is a critical component of pharma and biopharma manufacturing: an imprecise or false positive result can result in quarantine of a batch and repeat sample analysis which costs both time and materials.
Although numerous samples are collected throughout a drug manufacturing process, sampling technologies must:
- Not compromise the process: Samples should be taken according to validated procedures that do not introduce contamination to the process or otherwise affect process integrity.
- Collect representative samples: Samples collected must accurately represent the characteristics of the material being sampled.
- Be time and cost effective: to avoid being a burden on process economics.
- Be operator friendly: to avoid errors due to operator inexperience and avoid safety issues due to product exposure.
For additional information on sampling solutions, please visit Bioprocess Sampling Systems: Find the Right Sampling Approach for Your Needs.
