Understanding Listeria: Guide to Listeria Monocytogenes and Its Impact on Food Safety
What is Listeria? A Look at the Genus and Its Species
Listeria is a genus of Gram-positive, non-spore forming, rod-shaped bacteria. Among the known species, Listeria monocytogenes stands out as a significant human and animal pathogen. Other species, such as Listeria ivanovii, primarily affect animals, while the remaining species are generally considered harmless environmental bacteria.
Listeria bacteria are notable for their ability to thrive at refrigeration temperatures (2–8 °C), which poses a serious risk of contamination in ready-to-eat foods. Due to this risk, many countries have implemented strict food legislation that specifies limits for Listeria spp. and Listeria monocytogenes in food products.
Section Overview
- The Nature of Listeria Monocytogenes: Habitat and Risks
- The Importance of Testing for Listeria in Food Safety
- What is Listeriosis? Understanding the Infection
- Why Are Listeria Infections on the Rise?
- Biochemical, Rapid and Culture Methods for Detecting Listeria monocytogenes
- Listeria monocytogenes Testing workflow Guidelines
- Products for Listeria Testing in Food and Environmental Samples
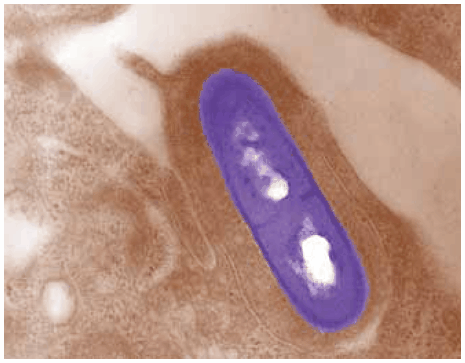
Figure 1.Electron micrograph of a Listeria monocytogenes bacterium in tissue.
Listeria monocytogenes exists ubiquitously in plants, soil and the guts of birds, fish, shellfish and some mammals, including humans. Studies suggest that 1-10% of humans may be intestinal carriers of L. monocytogenes. Foods that pose special risks include:
- Raw or processed meat
- Raw milk products
- Raw or smoked fish
- Ready-prepared salads
- Long-stored vacuum-packed foods
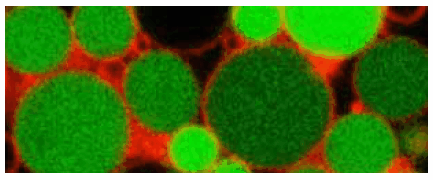
Figure 2.L-form of Listeria monocytogenes (source: M. Loessner and Y. Briers, ETH Zurich)
In food safety practices, it is crucial to conduct risk-related quality controls that include testing for L. monocytogenes and the broader Listeria genus. The presence of Listeria, particularly L. innocua, serves as an indicator of critical hygienic conditions during food production.
The incidence of foodborne infections caused by Listeria has surged, leading to an increased demand for reliable and rapid detection methods. Traditional culture methods are being supplemented by immunological techniques, which offer superior specificity and sensitivity.
What is Listeriosis? Understanding the Infection
Listeriosis is an infection caused by Listeria monocytogenes, recognized as one of the most widely distributed foodborne pathogens globally. It poses severe health risks, particularly to immunocompromised individuals, pregnant women, and neonates. Despite the relatively low number of cases, the mortality rate among infected individuals is alarmingly high.
Listeriosis can manifest in various ways, ranging from mild gastroenteritis to severe conditions such as sepsis, meningitis, and encephalitis. Symptoms may include:
- Flu-like symptoms
- Fever and muscle aches
- Gastrointestinal issues (nausea, diarrhea)
- Neurological symptoms (headache, stiff neck, confusion)
In pregnant women, listeriosis can lead to serious complications, including miscarriage, stillbirth, premature delivery, or infection in the newborn. A lot of research was done in recent years concerning the mechanism which this pathogen is using to invade the host and was found that L. monocytogenes is replicated rapidly in the cytosol of host cells like macrophages and lymphocytes(4).
Why Are Listeria Infections on the Rise?
Recent trends in food consumption patterns and the demand for longer shelf life have contributed to the increased incidence of listeriosis. The growing variety of “ready-to-eat” and “ready-to-cook” food products, along with longer storage at cool temperatures (4–8 °C) are some of the reasons for such problems. Innovative food preparation technologies, such as Cook Chill and sous-vide, while beneficial for extending shelf life, have also led to increased challenges in managing Listeria risks.
Statistics on Listeriosis in the United States
Listeria monocytogenes is the infectious agent responsible for the foodborne illness Listeriosis. In the United States, an estimated 2,500 persons become seriously ill with listeriosis each year. Of these, 500 die. (source: Dr. Balasubr Swaminathan; Peggy Hayes; CDC - Division of Bacterial and Mycotic Diseases: Listeriosis, 2002)
Listeria species are killed by heating steps, but the bacterium is relatively insensitive to high concentration of salts and acids. It also can multiply at fridge temperatures and inside vacuum packaging.
Biochemical Tests
The biochemical profile of Listeria includes several key characteristics: Catalase positive, oxidase negative, and ferments carbohydrates to produce acid without gas. Additionally, Listeria exhibits hydrolysis of esculin and sodium hippurate, is methyl red positive, and produces ammonia from arginine. It shows a negative reaction for hydrogen sulfide production, is indole negative, nitrate reductase negative, and does not liquefy gelatin, hydrolyze starch, or hydrolyze urea.
Further differentiation of Listeria species, particularly L. monocytogenes, can be achieved through additional biochemical tests. The process begins with the β-hemolysis test, where L. monocytogenes tests positive. This is followed by assessing carbohydrate fermentation abilities, such as positive results for rhamnose and methyl α-D-mannopyranoside fermentation, along with a positive CAMP test. In the CAMP test, certain Listeria species can enhance the hemolysis of Staphylococcus aureus. More information about this method can be found in the Rhamnose Broth data sheet. Refer the below table for biochemical testing products.
Biochemical Testing Products.
Another approach for phenotypic identification involves testing the fermentation capabilities of rhamnose, xylose, and mannitol. For a visual guide, refer to the identification flow chart in Figure 3.
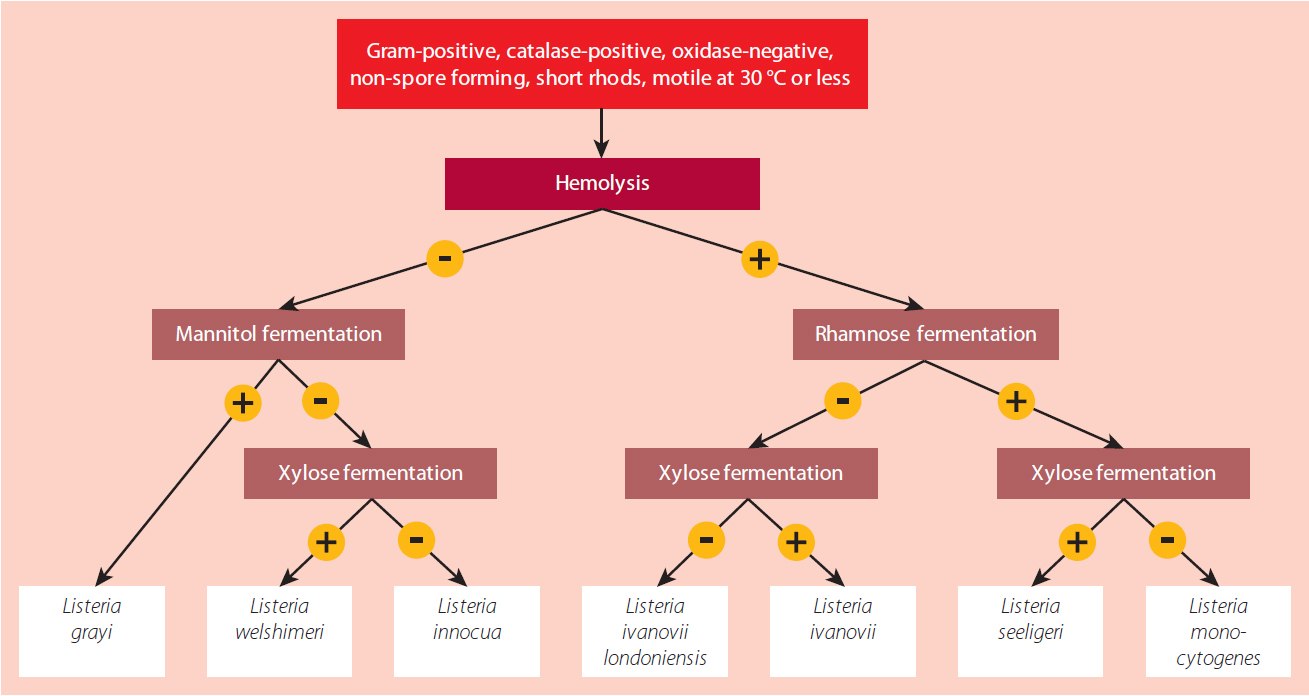
Figure 3.Schematic of biochemical identification for Listeria spp. based on carbohydrate fermentation tests and hemolysis (Source: Handbook of Listeria monocytogenes, 2008)
Culture Method using Chromogenic Media
An interesting and effective solution for confirming L. monocytogenes is the use of chromogenic media. Various commercially available chromogenic media exist, with the most notable being Listeria agar according to Ottavani and Agosti. This agar is described as horizontal method in EN ISO 11290:2017 (parts 1 and 2) to be used for detection and enumeration of Listeria monocytogenes and Listeria spp. in the food chain. For more information on these standards, visit our dedicated page: Standards for Detection and Enumeration of Listeria monocytogenes and Listeria spp. in the food chain
This agar employs specific systems for differentiation:
- Detection of β-glucosidase activity: This is achieved by including X-glu (5-bromo-4-chloro-3-indolyl-β-D-glucopyranoside) as a chromogenic substrate. This enzyme is present in all Listeria species, resulting in blue-green colonies on the agar.
- Detection of pathogenic Listeria: Lecithin, a phosphatidylinositol-specific phospholipase C, is incorporated into the selective media. Listeria monocytogenes hydrolyzes the specific purified substrate added to the agar, producing an opaque halo around the blue-green colonies. Most Listeria ivanovii also produce an opaque halo around colonies after 48 hours. In contrast, non-pathogenic Listeria species, such as Listeria innocua, only show blue-green colonies.
For more details on the detection systems used in chromogenic and other confirmation media, refer to Table 1c. To enhance the selectivity of the media, phenyl ethanol, along with high concentrations of lithium chloride and sodium chloride, is added. Additionally, antibiotics such as moxolactam, nalidixic acid, polymyxin B sulfate, ceftazidime, amphotericin B, acriflavine, cycloheximide, colistin sulfate, cefotetan, and fosfomycin are included to inhibit the growth of fungi, Gram-negative, and Gram-positive bacteria.
Rapid Methods for Listeria Testing
In addition to classical methods, we can also offer a range of rapid methods which also meet various regulatory requirements.
Among our offerings are immunological methods, such as the Assurance® EIA and TRANSIA® PLATE screens. The Assurance® EIA (Enzyme Immunoassay) utilizes specific antibodies to detect Listeria antigens in food samples, providing reliable results in a shorter time compared to traditional culture methods. Similarly, the TRANSIA® PLATE screens employ an enzyme-linked immunosorbent assay (ELISA), allowing for direct and efficient testing of multiple samples simultaneously.
Our rapid diagnostic offerings include Singlepath® Listeria monocytogenes and VIP® Gold for Listeria, both designed for efficient pathogen detection in food samples. Singlepath® Listeria monocytogenes is a lateral flow immunochromatographic test utilizing gold-labeled antibodies to detect L. monocytogenes antigens, delivering reliable results in just 20 minutes without the need for additional equipment. Similarly, VIP® Gold for Listeria is a single-step visual immunoassay that combines convenience and accuracy, featuring a built-in control reaction for optimized pathogen testing with enrichment media.
We also provide cutting-edge molecular methods, including the Assurance GDS® system (Genetic Detection System). The GDS system leverages polymerase chain reaction (PCR) technology to amplify and detect Listeria DNA in food samples, ensuring high sensitivity and specificity.

For testing the performance of media and assays, using Vitroids™ and LENTICULE® discs offers a time-saving solution by eliminating the need for preparing stock cultures. These innovative products require no recovery time or pre-enrichment steps, streamlining the testing process. The concentrations of the organisms are specifically designed to minimize or eliminate the need for dilutions, making them user-friendly. The discs dissolve easily in liquid media and automatically on agar plates, ensuring straightforward handling and a cost-effective solution.
Recent advancements in technology have led to significant improvements in the field of Microbiological Reference Materials. Key areas of development include enhanced stability, improved temperature resistance, precise adjustments to the defined CFU range, reduced rehydration time, and better reproducibility within batches. Moreover, each disc is produced in compliance with ISO 17034 standards for reference material production and is certified according to ISO/IEC 17025, ensuring high-quality and reliable results.
For more information on Vitroids™ and LENTICULE® discs, please visit our dedicated page.
Through ongoing innovation, we have developed reliable media and biochemical tests for Listeria in compliance with EN/ISO methodologies. These media are specifically designed to promote the selective growth and identification of Listeria in food products, ensuring quick and reliable results.
Detailed information about common media, tests, and related products can be found in can be found in tables given below.
Plating out Identification Media:
Confirmation Media (for Differentiation):
Listeria monocytogenes Testing workflow Guidelines
Listeria is a widespread microorganism found in various environments, and Listeria monocytogenes is a particularly harmful pathogen known to cause severe foodborne illnesses and infections. Therefore, it is essential to conduct rigorous testing of food products and laboratory environments for Listeria monocytogenes. The testing process is regulated by international standards to ensure consumer safety.
How can you stay compliant with these regulatory standards while achieving the most accurate results? In this video, you will be guided through the Listeria monocytogenes testing workflow based on ISO 11290, FDA-BAM, and USDA-FSIS guidelines. You will also discover our solutions designed to help you maintain compliance and enhance testing accuracy.
20-minute quick learning: detection of Listeria monocytogenes according to international standards
Presenter: Dr. Andreas Bubert, Merck KGaA, Darmstadt, Germany
Dr. Bubert brings over 25 years of experience in the food and beverage industry and has authored more than 40 publications.
Broth Media
Supplements for Broth and Agar Media
References
To continue reading please sign in or create an account.
Don't Have An Account?