Analysis of PFAS in Food Packaging Materials Using ASE, SPE and LC-MS/MS
Abstract
A liquid chromatography-tandem mass spectrometry (LC-MS/MS) method was developed for the analysis of per- and polyfluoroalkyl substances (PFAS) in food packaging materials, including compostable bowls, microwave popcorn bags, grease-proof paper sheets, and french fries paper bags. Samples were cut into small pieces and mixed with diatomaceous earth. After addition of isotopically labeled internal standards, target PFAS analytes were extracted using methanol by accelerated solvent extraction (ASE) method. The extracts were purified using Supelclean™ ENVI-WAX™ SPE cartridges to minimize matrix interferences. Analysis was performed using Ascentis® Express PFAS delay column and analytical column. Limits of quantification (LOQs) for all 25 PFAS compounds were in the range of 0.1-1.0 ng/g and recoveries obtained for the sample matrices spiked at 50 ng/g ranged from 55.4 to 130.4%.
Section Overview
Introduction
Per- and polyfluoroalkyl substances (PFAS) are widely used around the world, encompassing over 4,700 individual compounds.1,2 They are valued for their hydrophobic and oleophobic properties and are commonly incorporated into various consumer products, including disposable food packaging, cookware, outdoor gear, furniture, and carpets.3 However, PFAS are highly persistent, and many compounds are mobile, bio accumulative, and/or toxic.4,5 As a result, the presence of PFAS residues in food and the environment has garnered increasing scrutiny from the regulatory agencies. The United States Environmental Protection Agency (US EPA) established EPA 1633 method for determination of 40 PFAS analytes in aqueous, solid, biosolids, and tissue samples.6 Similarly, the Chinese national food safety standard GB 31604.35 2016 was introduced to evaluate PFOS and PFOA levels in food contact materials and related products.7
In the present study, sample preparation strategies described in EPA 1633 and GB 31604.35-20168 were combined to develop a liquid chromatography/tandem mass spectrometry (LC-MS/MS) method for determining 25 PFAS analytes in food packaging materials, including compostable bowls, microwave popcorn bags, grease-proof paper sheets, and french fries paper bags (Figure 1). For this assessment, a single isotopically labeled internal standard (PFOA 13C4) was used.




Figure 1. Food packaging material samples investigated in this work.
Experimental
Based on procedures described in GB 31604.35-2016 and LC-MS/MS conditions specified in EPA 1633, packaging materials samples were first subjected to accelerated solvent extraction (ASE). The resulting extracts were then cleaned and concentrated by solid phase extraction (SPE) using Supelclean™ ENVI-WAX™ SPE cartridges on a PTFE free Visiprep™ SPE vacuum manifold was performed. The purified extracts were finally analyzed by LC-MS/MS using superficially porous particle (SPP) Ascentis® Express PFAS analytical and delay columns.
Internal Standard Preparation
- Isotopically labeled internal standard stock solution (1 μg/mL): A perfluorooctanoic acid-13C4 solution (PFOA-13C4) in methanol was used without further treatment.
- Isotopically labeled internal standard solution (IS, 400 ng/mL): 400 μL of the internal standard stock solution was transferred into a 1.5 mL centrifuge tube 600 μL methanol was added.
Standard Preparation
- Single PFAS analytes stock solutions (25 in total): Individual PFAS solutions (100 μg/mL) were prepared by dissolving an appropriate amount of each PFAS reference material in a measuring flask containing 10-20 mL methanol.
- PFAS mixed analytes working solution (PFAS WS): 10 μL of each of the 25 single PFAS stock solutions was pipetted into a 1.5 mL centrifuge tube and 750 μL methanol was added. The concentration of each of the PFAS analytes in the resulting solution was 1 μg/mL.
- Mixed standard solutions (PFAS SS): 500, 200, 100, 50, 20, 10, and 5 μL of PFAS WS were transferred into seven individual 1.5 mL centrifuge tubes and diluted to 1 mL with methanol. The concentration of each PFAS in the resulting solutions (standards #1 to #7) was 500, 200, 100, 50, 20, 10, and 5 ng/mL.
10 μL of standards #2, #3, #4, #5 and #6 were transferred into five individual 5 mL centrifuge tubes and diluted to 1 mL with methanol to obtain an additional set of PFAS standard solutions (standards #8 to #12) with PFAS concentrations of 2, 1, 0.5, 0.2, and 0.1 ng/mL, respectively. - 1 mL of each PFAS SS (#1 - #12) were transferred separately into 2 mL glass vials, 50 μL of internal standard solution (IS) was added, and the solutions were mixed thoroughly.
Sample Preparation
Food packaging materials were cut into pieces measuring 0.5 cm x 0.5 cm using a paper cutting machine.
- 1 g of sample was weighed into a 200 mL glass beaker. For method recovery determination, 50 μL of PFAS WS (spiking level: 50 ng/g) was added and the following preparation procedure was performed.
- 6 g diatomaceous earth was added, and the mixture was stirred thoroughly with a spoon.
- A filter membrane and sieve plate were placed at the bottom of a 34 mL ASE extraction tube, and the bottom screw cap was tightened.
- The mixture prepared in step 3 was poured into the extraction tube using the funnel supplied with the ASE instrument.
- 50 μL IS was added to the extraction tube.
- A filter membrane was placed on top of the extraction tube, and the top screw cap was tightened.
- The tube was placed in the ASE oven, and the extraction process was initiated. Extraction conditions are provided in Table 1.
- The extract was collected in a 100 mL glass bottle, and a final volume of 50 to 70 mL was obtained.
- The extract was evaporated to ~ 0.5 mL under nitrogen at 40 °C.
- 10 mL of water was added to the extract, and the solution was shaken well.
- The solution was transferred to an SPE tube for clean-up following the procedure described in Table 2.
LC-MS/MS Analysis
Standards and samples were analyzed by LC-MS under the instrumental conditions described in Tables 3 to 5.
Table 5. MRM transitions used for analyte detection

Figure 2.Pictures of samples dried with nitrogen after ASE (“1-2”: microwave popcorn bag sample; “2-4”: compostable bowl sample; “3-6”: grease-proof paper sheet sample; “4-8”: french-fries paper bag sample).
The extracts obtained after ASE were too contaminated/matrix loaded for direct injection onto the HPLC column, as doing so could result in clogging or column damage and reduced column lifespan. Therefore, an SPE cleanup was carried out to purify the sample extracts. However, increasing the number of sample preparation steps typically leads to lower recovery rates. If higher recovery is desired, omission of the SPE step may be considered, although potential matrix interference in PFAS determination and its impact on column lifetime would need to be evaluated.
After cleanup, the samples were analyzed by LC-MS using dedicated superficially porous particle (SPP) columns for PFAS, namely Ascentis® Express PFAS 2.7 µm delay and analytical columns. The chromatographic separation of a standard mixture containing 25 PFAS compounds at 10 ng/mL each is shown in Figure 3, and the corresponding retention times and calibration data are summarized in Table 6. Within their specified calibration ranges, the square of linear regression coefficient values R2 were greater than 0.98 for all analytes.

Figure 3.LC-MS/MS chromatogram of a standard mixture of 25 PFAS (10 ng/mL each).
Table 6. Retention time and calibration data
The chromatogram obtained from analysis of a french-fries paper bag spiked with PFAS at 50 ng/g for each compound is presented in Figure 4, and the respective recovery data and limits of quantification (LOQs) for all matrices are provided in Tables 7 and 8.
Results obtained for the compostable bowl sample showed recoveries for all 25 PFAS compounds in the range of 55.4 – 104.6%, with LOQs between 0.1 – 1.0 ng/g. For microwave popcorn bags samples, the recoveries were between 59.5% and 118.1% and LOQs ranged from 0.1 to 1.0 ng/g. For grease-proof paper sheet samples, the recoveries were found to be in the range of 62.1 – 111.0% and LOQs between 0.1 to 1.0 ng/g. For french fries bag samples, the recoveries were within the range of 67.7 – 130.4% and LOQs were 0.1 – 1.0 ng/g. According to GB 31604.35-2016, the required limit of detection (LOD) is 1.0 ng/g and the required LOQ is 2.0 ng/g for PFOS and PFOA in food packaging materials. The developed method therefore meets these requirements of the GB 3164.35-2016.
The target LOQs specified in AOAC SMPR® 2025.001 (Standard Method Performance Requirements (SMPRs®) for Per- and Polyfluoroalkyl Substances (PFAS) in Food Packaging Materials)9 are defined as 20 µg/kg for individual analytes and the developed method also fulfills this requirement. The recovery criterion of 60-115% for samples at or above 10 µg per kg was satisfied for the majority of investigated analytes.
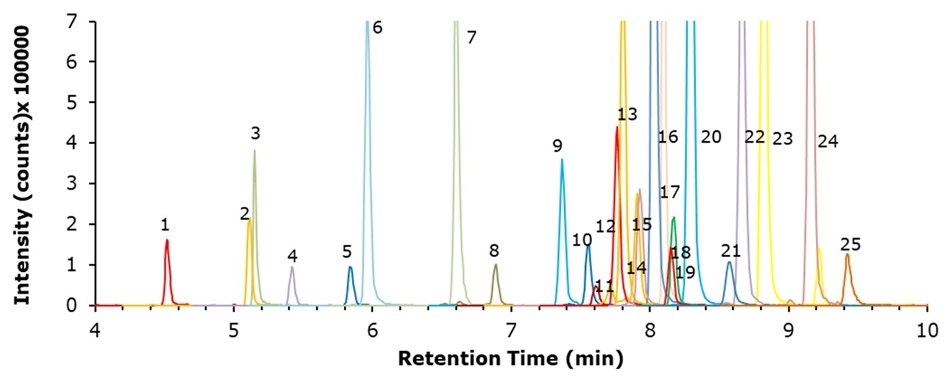
Figure 4.LC-MS/MS chromatogram obtained for the analysis of a french-fries paper bag spiked at 50 ng/g of each PFAS.
Table 7. Results for compostable bowl and microwave popcorn bag samples
Average method recovery (n=3) and LOQs for the 25 PFAS spiked at 50 ng/g
Table 8. Results for grease-proof paper sheet and french-fries paper bag samples
Method recovery and LOQs for the 25 PFAS spiked at 50 ng/g
For all four investigated samples, background levels of the 25 analytes in unspiked samples were below the compound specific limits of quantification (LOQs).
Method Considerations
Solvent Effect
Methanol was used as the sample diluent for the final test solution. However, the initial mobile phase exhibited a significantly weaker elution strength (A:B = 98:2, where [A] is 2 mM ammonium acetate in a 5:95 acetonitrile/water mixture and [B] consisted of acetonitrile). This disparity in elution strengths is known to negatively affect the shape of chromatographic peaks. For certain PFAS analytes with relatively high LOQs, the injection volume was increased to 5-10 μL for enhanced signal response, but this resulted in asymmetric and deteriorated peaks. When the injection volume was set to 2 μL, no such negative effects were observed and satisfactory LOQs were maintained.
Method Recovery
Accelerated solvent extraction (ASE) and solid-phase extraction (SPE) were employed for sample extraction and cleanup. To achieve optimal recovery rates with ASE, samples were cut into small pieces and combined with diatomaceous earth to increase the contact surface area with the extraction solvent. However, if the sample and diatomaceous earth mixing was insufficient, the paper samples tended to agglomerate under high-pressure extraction conditions, resulting in decreased recovery rates. The low recovery observed for certain PFAS analytes was therefore attributed largely to uneven mixing. Enhancement of PFAS recovery during ASE may require additional research, including the use of smaller sample pieces or reduction of extraction pressure.
Internal Standard Used
Recovery is also influenced by the selected internal standard. EPA 1633 recommends the use of multiple internal standards covering a wide range of compounds. In the present study, only a single IS compound (PFOA-13C4) was used for economic reasons. More accurate quantification and improved recovery may be obtained using multiple isotopically labeled internal standards to better compensate for matrix effects in the mass spectrometer and sample loss during the sample preparation process. Further investigation is required to evaluate potential improvements in overall method performance and applicability.
Conclusion
An LC-MS/MS method was developed for the determination of 25 PFAS compounds in food packaging materials using accelerated solvent extraction (ASE) followed by SPE cleanup. The investigated samples included compostable bowls, microwave popcorn bags, grease-proof paper sheets, and french fries paper bags. Samples were cut into small pieces, extracted by ASE, and further purified by SPE, using a Supelclean™ ENVI-WAX™ cartridge to reduce matrix interference. The limits of quantification and method recovery were evaluated against an external calibration and using a single internal standard (PFOA 13C4) for all measurements. All linear regression coefficients (R²) within their respective calibration ranges were greater than 0.98, indicating suitability of the LC method for determination of the 25 PFAS analytes. For all four sample matrices, LOQs ranged from 0.1-1.0 ng/g for all compounds. Recovery rates were assessed by spiking blank samples at ~ 50 times the LOQ (50 ng/g) and recoveries ranged from 55.4 to 130.4%.
Future work may include evaluation of a broader range of internal standards to further improve method performance and applicability to additional PFAS compounds.
Explore our complete PFAS portfolio to streamline method development, meet evolving regulatory requirements, and achieve confident trace-level detection across complex matrices. Browse products, download application resources, and find the right solutions for your workflow today at PFAS Testing.
See our complete offering of PFAS reference materials.
Related Products
HPLC and Sample Preparation
Solvents, Reagents, and Accessories
Reference Materials
References
To continue reading please sign in or create an account.
Don't Have An Account?