Conferma® ELISAs: Pursuing a “Pharmagrade” Immunoassay
Biomarker research requires reproducible detection over time. The Conferma® ELISAs are developed and extensively verified to fulfill this requirement.
Section Overview
Pharmagrade Immunoassays
The need for improved immunoassay assay performance has long been discussed. A white paper published in Bioanalysis by Khan et al. in 20151 coined the term “Pharmagrade” to describe “kits with key characteristics, such as high inter-lot consistency, access to bulk reagents, analytical component documentation and standardized validation practices” that “will increase reproducibility and decrease the need for extensive end-user work.”
Following this train of thought, our Conferma® ELISA program was started. We also integrated concepts from more recent publications including the ICH-M10 guidance and the CPath Institute's “Points to Consider” document (Piccoli & Sauer, 2019). While explicitly not for RUO (Research Use Only) ELISAs, we decided to explore assays that could fit the spirit of these documents, and as a result each ELISA follows the following key principles:
- Physicochemical validated critical reagents (lot-to-lot)
- Component level trend charting
- Native sample library-based lot release testing
Through this we hoped to achieve “Pharmagrade” off-the-shelf commercial ELISAs. In this article we demonstrate the real-world impact of some of the principles we have adopted.
Conferma® ELISA Development
Conferma® ELISAs are developed using in-house critical reagents (antibodies and standards) put through an advanced screening and validation process to evaluate affinity (mAbs) and endogenous similarity (standard) before creating finished ELISAs that are tested and batch released using native samples. The process leads to assays with a high level of sample detectability and reproducibility (Figure 1).
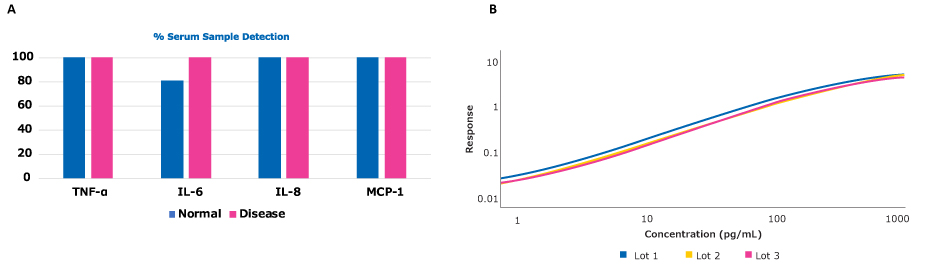
Figure 1.Conferma® ELISAs provide high sample detection and lot-to-lot consistency. (A.) The % sample detection (above limit of detection, or LOD) in normal and diseased serum samples using Conferma® ELISAs for the analytes TNF-α, IL-6, IL-8, and MCP-1. (B.) Example of finished kit testing of the TNF-α ELISA. Calibration curves from 3 lots were compared for mathematical similarity where a value of 1 is considered perfect parallelism. Lots 1, 2, and 3, are represented by the blue, yellow, and magenta lines respectively. Parallelism of Lot 2 vs. Lot 1 was 0.983 and Lot 3 vs. Lot 1 was 0.992.
Critical Reagent Testing
Our component level testing looks to establish the exact nature of the calibrator materials (standards and controls), and its relationship with the antibodies that create the assay. Our standard tests include:
- AAA: Amino Acid Analysis, to normalize reagent concentration
- Densitometry (Purity)
- Mass Spectrometry (LC-MS/MS and RP-LC-UV-MS)
- Surface Plasmon Resonance (SPR)
The Mass Spectrometry techniques provide a predicted sequence (Figure 2) and mass which can be qualified lot to lot and assessed (in the case of the standard) against the UniProt database to ensure similarity to the native target molecule. SPR confirms that the affinities of the monoclonal antibodies have remained strong and consistent as reagent lots change.

Figure 2.LC-MS/MS Analysis. Sequence coverage of two lots of IL-6 Conferma® ELISA calibrator material based on LC-MS analysis. The blue highlight shows the identified sequences of IL-6.
These tests allow us to confirm reagent similarity, evaluate the consistency of their relationships, and provide data presented in lot-specific certificates of analysis which allow users to keep a record of the critical reagents driving their research (Table 1).
Assay Testing: Verifying Performance
Assay verification testing determines the operating parameters to which an assay is expected to perform both in-house and in your laboratory. The influence of these tests may be defined by the work being undertaken; for instance, whether the kit’s sensitivity, range, and consistency adequately describe the change in your biomarker over the course of a study. Recommended tests are well-documented2-5, and we include many when testing Conferma® ELISAs to give as comprehensive a picture as possible. In Table 2 you can see the list of applied tests during verification; however, the guidance calls out three changes to recommended parameters, specifically.
- Sensitivity (Figure 3): sensitivity is often defined as the LOD (limit of detection, “x” SD above background); we instead use Lower Limit of Quantitation, an anchored value on the standard curve.
- Sample Parallelism (Figure 4): we use the back-calculated value of a native sample, rather than the linearity (which uses a recombinant protein).
- Cross-reactivity: the signal from the tested molecule should be less than 20% of the signal (O.D.) of the assay’s LLOQ.
These changes are adopted to increase the certainty that the assay is interpolating a unique signal that lands reproducibly on the assay curve.
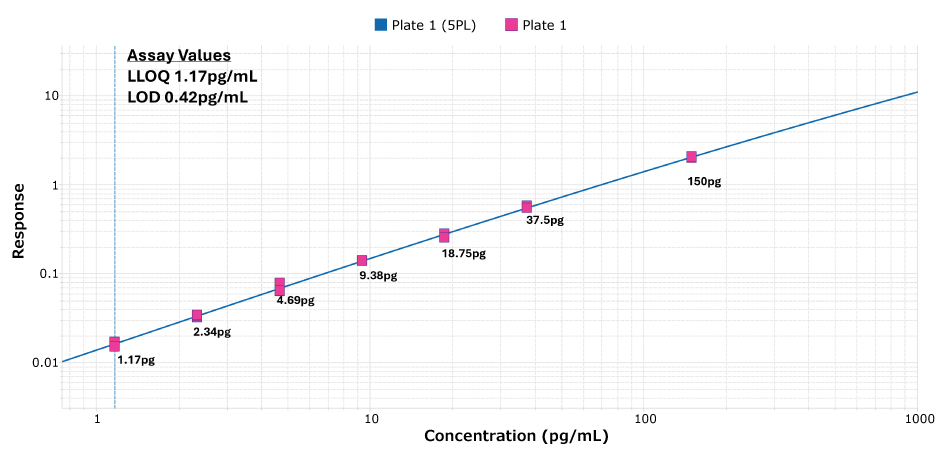
Figure 3.LLOQ example. The LLOQ is the lowest analyte concentration demonstrating acceptable precision (≤20%) and recovery (≤20%). The LLOQ of the Conferma® IL-6 ELISA is 1.17 pg/mL, the dotted line at lowest point in this standard curve.
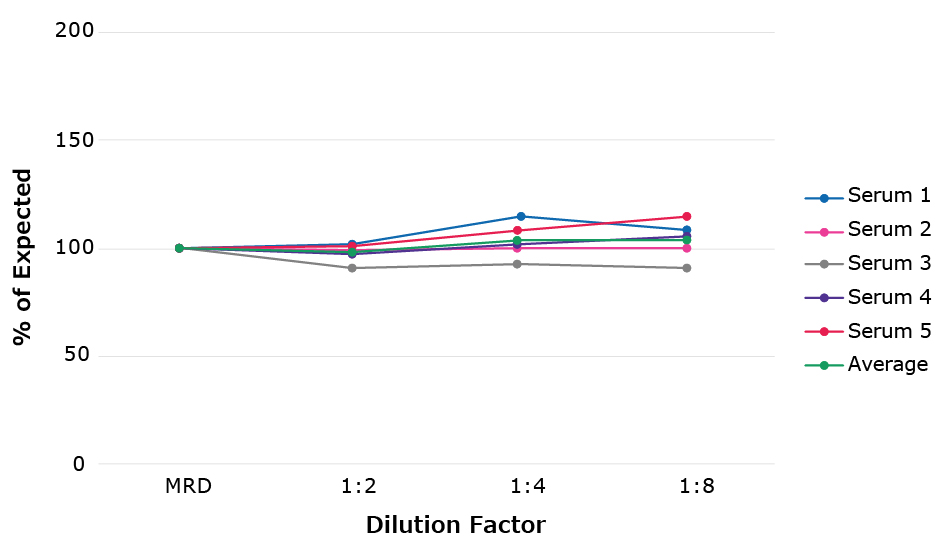
Figure 4.Sample parallelism example for the Conferma® MCP-1 ELISA (EZHMCP1-99KRM). Five endogenous serum samples with high levels of target protein were diluted and back calculated to the original value.
Assay Lot-to-Lot Consistency
Reproducibility is a key concern for most immunoassay users, both when repeating experiments or across longitudinal studies. Conferma® ELISAs build upon our existing quality practices:
- Establish a recombinant gold standard to test against every subsequent lot of standard
- Test each component individually within a full assay
- Use trend charts to analyze all historical data
- Perform finished lot sample library testing (specific to each Conferma® ELISA)
Finished kit sample testing has been added to ensure that each lot is bridged to the previous lot using a native sample library (serum and plasma, both un-spiked and spiked with the standard). The release criteria can be found on the CoA and is based on 80% of tested samples being within 15% or 20% of the prior value, which is further evaluated against the original value of the library. In Figure 5, you can see the library sample range and the %CV across 5 lots of TNF-α (EZHTNFA-150K) and IL-6 (EZIL6-98K) ELISAs. Across the Conferma® ELISA range, we calculate a mean CV <10% from sample library testing.
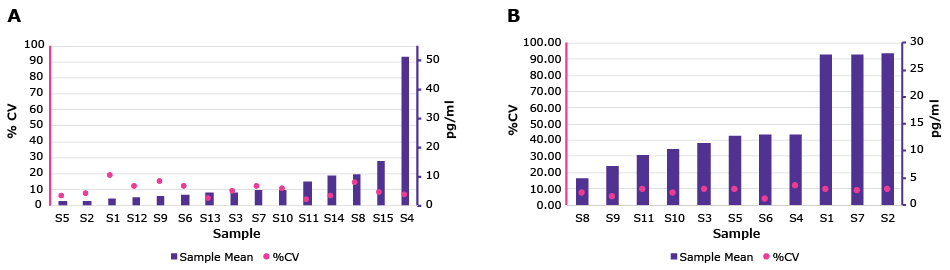
Figure 5.Five lots of (A) Il-6 and (B) TNF-α Conferma® ELISAs tested against their sample libraries.
Pharmagrade ELISAs
Conferma® assays were developed to put into practice the published thoughts of researchers on immunoassay verification and validation, with the pursuit of a “Pharmagrade” ELISA. Adopting a granular approach to reagent creation and evaluation leads to ELISAs which can pass extremely stringent finished kit QC criteria. As such, scientists can trust these products to work reliably every time. We hope that this process is a significant step toward the early vision presented by the authors who coined the term “Pharmagrade”.
Acronyms & Definitions
LOD: Limit of Detection (a given number of SD of the Blank wells, used to provide an estimate of where the signal may be considered the assay derived rather than background)
LLOQ: Lower Limit of Quantitation (where %CV & Recovery meet defined criteria on the curve above which all others meet the same criteria)
O.D.: Optical Density (the signal from a colorimetric assay, based on the absorbance of emitted light)
CV: Coefficient of Variance (% difference between two values)
QC: Quality Control
For Research Use Only. Not For Use In Diagnostic Procedures.
Associated Products
Belysa® immunoassay curve fitting software was designed to demonstrate immunoassay method consistency over the course of a study or through the long-term use of an assay, proving the ideal complement to the Conferma® products. Belysa® software is a multiplatform solution that works with ELISA, Luminex® multiplex, and SMC® assays. Find out how it is designed with method consistency in mind.
