New Grafted Zwitterionic HILIC Stationary Phase for Glycan Profiling
Abstract
This investigation assessed the separation efficacy of zwitterionic chromatography columns designed for hydrophilic interaction liquid chromatography (HILIC) in glycan profiling of immunoglobulin G (IgG)-derived glycans. Glycans were enzymatically released from IgG samples and analyzed in their reduced forms using high-performance liquid chromatography coupled with electrospray ionization high-resolution mass spectrometry (HPLC-ESI-QTOF-MS/MS). The comparative analysis focused on glycan retention characteristics, peak properties, and overall resolution. The columns featured stationary phases with sulfobetaine (SB) and phosphorylcholine (PC) functionalities, including a final prototype of the latter stationary phase applied to superficially porous particles (SPP, commercially introduced as Fused-Core® in the Ascentis® Express ZIC®-cHILIC). All three columns effectively produced informative glycan profiles, detecting and separating predominant glycan species. However, significant differences in retention and resolution were noted, with PC-modified columns showing superior separation, particularly between sialylated and non-sialylated glycans. This enhanced resolution is crucial, especially when mass spectrometry is not used. Additionally, the column with Fused-Core® particles achieved shorter analysis times with maintained or improved glycan resolution compared to its fully porous counterpart(s).
Section Overview
Introduction
Immunoglobulin G (IgG) monoclonal antibodies (mAbs) are crucial biotherapeutics requiring stringent quality control and characterization, particularly regarding post-translational modifications like glycosylation. IgGs feature a conserved N-glycosylation site in the Fc region and additional sites in the Fab region, resulting in complex biantennary glycans with approximately 30 variants identified in human serum. These glycans, composed of mannose, N-acetylglucosamine, galactose, sialic acid, and fucose, significantly influence IgG characteristics, including serum half-life, bioactivity, stability, and immunogenicity. Sialic acids play a key role in anti-inflammatory responses, while core fucosylation affects antibody-dependent cellular cytotoxicity (ADCC). The specific glycosylation profile is influenced by protein expression conditions and downstream processing, making it a critical quality attribute for IgG mAbs.
Biopharmaceutical glycan analysis typically involves enzymatic release followed by capillary electrophoresis or liquid chromatography, with fluorescent labelling being a predominant method for quality control. Mass spectrometry (MS) provides detailed qualitative and quantitative glycan assessments with MS/MS fragmentation aiding in glycan identification. Liquid chromatography techniques, particularly hydrophilic interaction liquid chromatography (HILIC), are effective for retaining and separating polar and charged compounds, with various stationary phases employed, including zwitterionic sulfobetaine (SB) and phosphorylcholine (PC) functionalities. An interlaboratory study led by National Institute of Standards and Technology (NIST) highlighted significant variability in glycosylation analysis results, underscoring the need for harmonized analytical methods.1 There is a demand for robust techniques that allow sensitive analysis across a wide dynamic range.
In this work, the glycan profile of IgGs from various sources obtained in HILIC-LC-MS/MS mode analysis on commercially available and prototype zwitterionic chromatography columns were evaluated and compared, focusing on retention and resolution. Column comparisons for additional HILIC stationary phases including surface-grafted stationary phases with SB and PC functionalities, utilizing Fused-Core® (superficially porous, SPP) and fully porous (FPP) particles (Figure 1) of different sizes including three columns having the same particle size and porosity but differing in their synthesis methods, and hence resulting in variations in polymer network and internal structure, is reported elsewhere.2
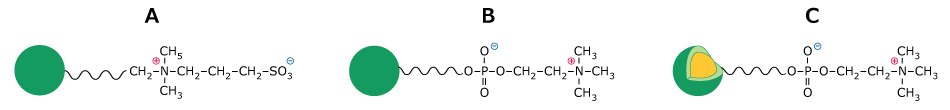
Figure 1.Sulfobetaine (SB) and phosphorylcholine (PC) functionalization on FPP and SPP.
Experimental
Chemicals and Reagents
The column evaluation utilised commercially available IgG standards of different origins (IgG human, IgG goat, IgG rabbit) as test solutes and PNGaseF derived from Elizabethkingia meningoseptica for glycan release. These were all sourced from Merck KGaA Darmstadt, Germany. All chemicals utilized in the study were of analytical or mass spectrometry grade. Water was generated in-house (Milli-Q® system).
Sample Preparation
The IgG standards for glycan release were prepared by dissolving them to a concentration of 2.5 mg/mL in a 50 mM ammonium bicarbonate buffer, which was adjusted to pH 8.0 using formic acid. To release N-glycans, 100 µL of the IgG solution was treated with 10 units of PNGase F and incubated at 37 °C for 18 hours. Following incubation, ice-cold ethanol at a ratio of 4:1 (ethanol to sample) was added to precipitate the proteins, and the mixture was centrifuged at 14,000 g for 10 minutes. The supernatant, which contained the released N-glycans, was carefully collected and transferred to new sample tubes. The samples were then dried using a Speed Vac® concentrator, after which 20 µL of fresh 0.5 M sodium borohydride (NaBH4) in 50 mM sodium hydroxide (NaOH) was added to facilitate the reduction of N-glycans. The reaction was terminated after overnight incubation at room temperature by adding 5 µL of glacial acetic acid. Prior to analysis, the samples were diluted with 80 µL of acetonitrile resulting in a total volume of 105 µL.
LC Method
Three different UHPLC columns (150 x 2.1 mm I.D.) supplied by Merck were assessed in this study. Their properties are summarized in Table 1. The columns differ in both the type of particle and the surface functionality. Column C utilised was a prototype ZIC®-cHILIC column, commercially released as Ascentis® Express 160 Å ZIC®-cHILIC column.
Chromatographic conditions and mass spectrometry analysis
Released glycans were separated using analytical HPLC columns (150 x 2.1 mm I.D.) with various particle dimensions, types, and surface chemistries, as outlined in Table 1. Glycan analysis utilized an Agilent Infinity II HPLC system, equipped with a binary pump, degasser, autosampler, and column thermostat, coupled to a Bruker Impact II QTOF mass spectrometer. Chromatographic separation was achieved through gradient elution. The eluent composition and gradient elution profile for the column comparison in this study were not optimised for each column, but conditions used were those established with a commercially available fully porous silica-based zwitterionic SeQuant® ZIC®-HILIC column (Column A, Table 1), which had been previously utilized for glycan profiling in-house. For initial elution conditions development, ammonium formate was tested as a mobile phase modifier at two different pH levels (3 and 7) and three concentrations (5, 30, and 50 mM). The results indicated that an acidic pH yielded higher responses and improved peak shapes for the primary glycans (FA2, FA2G1, FA2G2). Additionally, increasing the concentration of ammonium formate enhanced peak sharpness and intensity; however, a concentration of 50 mM resulted in a rapid accumulation of salt in the mass spectrometry (MS) source. The optimal sensitivity and glycan profile on Column A were achieved under positive ionization mode, utilizing ammonium formate (30 mmol/L) and formic acid (0.05%) as eluent modifiers in gradient elution mode. This eluent composition and gradient conditions was subsequently applied to all further column comparisons. Eluent and gradient conditions are given in Table 2 and mass spectrometer method settings are given in Table 3.
Data Analysis
The raw data were analyzed using Compass DataAnalysis 5.3 from Bruker Daltonics, along with Skyline software developed by the MacCoss Lab. The NIST Glyco Mass Calculator3 was employed to determine the expected mass of the glycan species. Observed versus theoretical masses for the ion [M+H]+ for the main glycan species originating from IgG human are shown in Table 5-7. Fragmentation data obtained from MS/MS analysis provided additional support for glycan identification, including the fragment for hexose-N-acetylhexosamine (366.14 m/z) applicable to all glycans, as well as the fragment for hexose-N-acetylhexosamine-sialic acid (657.24 m/z) for the identification of sialylated glycans. Extracted ion chromatograms corresponding to the primary adducts observed, namely [M+2H]2+, [M+H+Na]2+, and [M+2Na]2+, were utilized to visualize the glycan profile. The relative retention of each glycan was calculated using glycan A24 as a reference (the structure of which is detailed in Table 5). The retention time window, meaning the difference in retention time from first to last eluting glycan was determined and compared. Additionally, parameters such as peak widths, peak symmetry, and chromatographic resolution between different glycan species were assessed and are summarized in Tables 4 to 8. The glycans included in Tables 5-7 were all included in the calculations of retention time window, average peak widths and symmetries.
Results and Discussion
Glycan Retention and Peak Properties
The glycan chromatographic profiles obtained from the LC-MS/MS analysis of glycans enzymatically released from human IgG were evaluated, focusing on retention, resolution, and peak characteristics. The glycan chromatographic profiles for Columns A-C show significant differences in glycan retention and some variation in peak widths and peak asymmetry, Table 4. The glycan profile of human IgG revealed a variety of hybrid and complex glycans4 (examples given in Tables 5-7), including non-fucosylated glycans (A2, A2G1, A2G2), biantennary structures (FA2, FA2G1, FA2G2), triantennary forms (FA2Bi, FA2BiG1, FA2BiG2), and sialylated glycans (FA2G1S1, FA2BiG1S1, FA2G2S1, FA2BiG2S1, FA2G2S2, and FA2BiG2S2) with good agreement between observed (measured) and expected (theoretical) mass, as shown for [M+H]+. The longest retention times are recorded for the fully porous particle ZIC®-cHILIC column (Column B, PC phase) where A2 elutes at 7.6 minutes compared to at 5.4 minutes for Column A, and at 4.2 minutes for Column C. Column B also has the broadest retention time window, approximately 11 minutes from the first to the last eluting glycan, in contrast to less than five minutes for the fully porous ZIC®-HILIC column (Column A, SB phase). Column C, which shares the same surface chemistry (PC) as Column B, demonstrates shorter retention times consistent with its superficially porous particle properties. Overall, the peaks observed across all tested columns are relatively narrow, although some variations exist. The widest average peak width of 0.24 min was observed for Column B, while the narrowest average peak widths were recorded for Column C which exhibited peak widths of 0.13 min, measured as Full Width at Half Maximum (FWHM). This result aligns with established knowledge that superficially porous particles generally provide superior peak shape due to their shorter channels, which reduce diffusion, and result in lower backpressure. The peak symmetry was not markedly different between columns and between glycans of different elution times. This is expected since all glycans are eluting during linear gradient elution.
Glycan Retention, Resolution and Effect of Sugar Moiety Differences
The columns utilized in this study feature two distinct zwitterionic functionalities: sulfobetaine (SB) functionality found in ZIC®-HILIC type columns and phosphorylcholine (PC) functionality present in ZIC®-cHILIC type columns, as detailed in Table 1. The retention of hydrophilic compounds such as glycans on both phases is expected to be multifactorial and to a large extent a result of hydrophilic partitioning. Optimized conditions rely on stationary phase chemistry, analyte characteristics and solvent composition. In the column comparison, the chromatographic profiles for IgG (human) glycans exhibited a similar elution order across most columns but, under the experimental conditions used, the PC columns generally exhibit stronger retention and higher resolution compared to the SB column, as shown by the achieved baseline resolution for several adjacent glycan pairs, Table 6 and 7. In contrast, the SB column (Column A) show considerably lower resolution, with baseline separation only achieved for two adjacent peak pairs, Table 5.

Figure 2.Representation of glycan structures.
In HILIC, larger glycans, which possess additional sugar moieties, typically exhibit longer retention times than smaller glycans, which is indeed observed for this example when comparing retention times for glycans A2, A2G1 and A2G2, on all columns, Table 5-7. However, the specific nature of the additional sugars and the glycan structure also influence retention. For example, sialylated glycans contain sialic acid, which is a strong acid (pKa = 2.6), imparting both a hydrophilic character and a negative charge to the glycan. The difference in the location of the positive and negatively charge moiety of the zwitterionic functionality adds another separation handle. Especially for these charged glycans for which an additional repulsive or attractive force can be in play due to differences in the outer (furthest away from the silica particles) part of the zwitterionic moiety, which is expected to be more easily accessible to the glycan itself. In SB, the outer moiety is negatively charged, while in PC it is positively charged, Figure 1. The highest resolution between sialic acid-containing glycans and their non-sialylated counterparts (pairs 4 and 5) is observed on columns with PC surface functionality. Column C, Figure 4, exhibits good resolution for sialic acid and galactose-containing glycans, with performance that is better or comparable to Column B (Figure 3A, Table 8).
The sugar moiety fucose, having a hydroxyl group less than sugars such as galactose, is expected to yield decreased hydrophilicity to the glycan. At the same time, the core fucosylation potentially yields additional complexity, as the structure of the glycan is also affected, which in turn could influence the glycan hydrophilicity or induce steric effects. When comparing the retention time of the glycan FA2 to A2G1, and FA2G1 to A2G2, indeed the fucosylated glycans have shorter retention times on all columns, suggesting that for a glycan with the same number of sugar moieties but where one glycan is fucosylated and the other instead has an additional galactose unit, the fucosylated glycan is less hydrophilic. This can also be seen from the resolution between fucosylated and non-fucosylated glycans (pairs 15 to 17, Table 8) being much lower than for glycans differing in one galactose unit (pairs 9 to 14). The most effective separation between fucosylated and non-fucosylated glycans is achieved on the PC type columns B and C.
High-resolution separations of sialylated versus non-sialylated glycans (pairs 4 and 5) and glycans with different number of galactose units (pairs 6 to 14) are achievable on the superficially porous particle (SPP) column with 160 Å pores, Column C, with a total retention time window for the glycan profile of less than six minutes, resulting in shorter elution times and thus reduced analysis times compared to fully porous particle (FPP) Column B for which the retention time window is approximately 11 minutes.
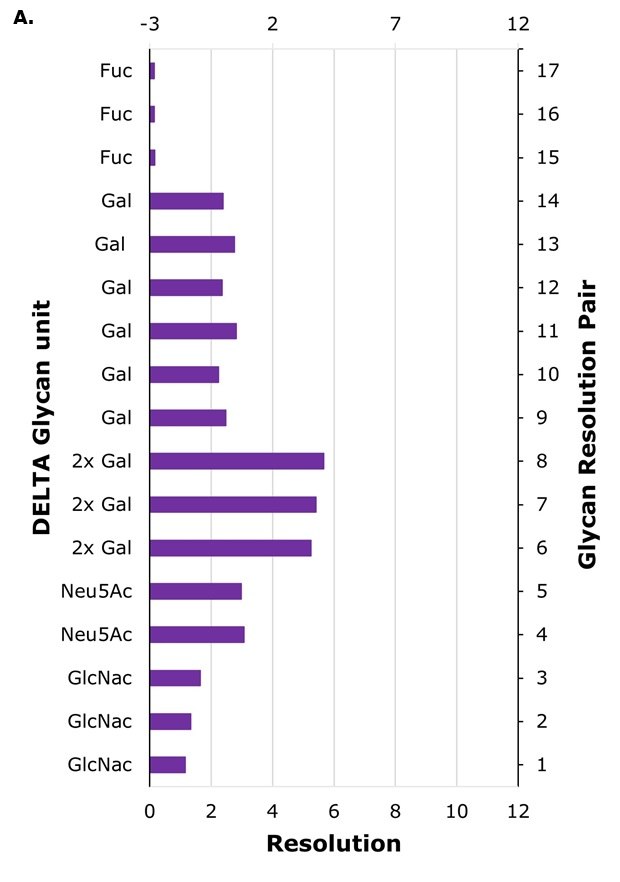
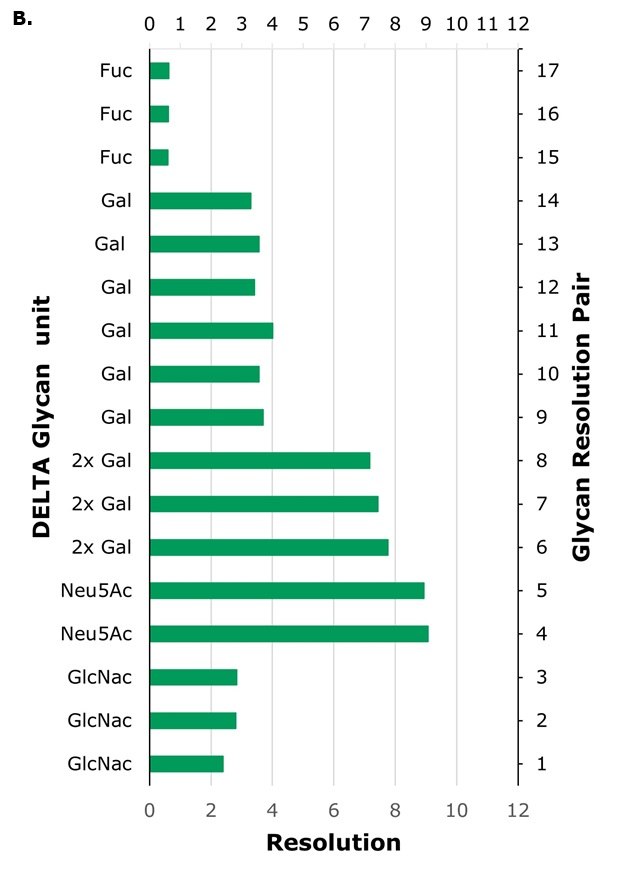
Figure 3. Glycan resolution on Column A (panel A) and Column B (panel B). Glycan pairs are given in Table 8.
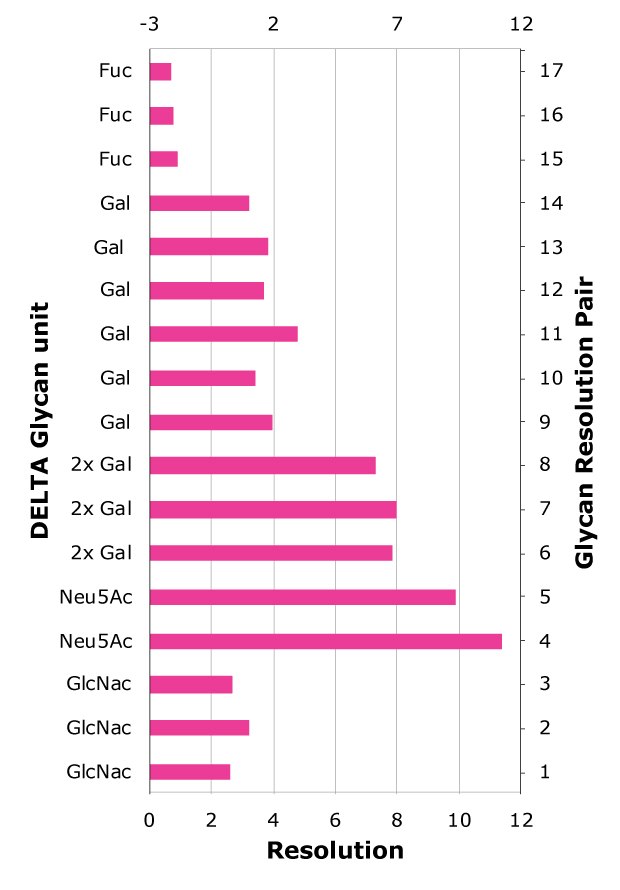
Figure 4. Glycan resolution on Column C. Glycan pairs are given in Table 8.
N-Glycan Profiles for IgGs of Different Origins
Based on the N-glycan profile observed for human IgG, Column C was identified as the optimal choice due to its ability to achieve high-resolution separations of closely related glycans and a retention time window of less than six minutes from the first to the last eluted glycan. This column was subsequently used to analyze the N-glycan chromatographic profiles of glycans released from rabbit and goat IgGs. As illustrated in Figure 5, the chromatographic profiles of N-glycans from human, rabbit, and goat IgGs exhibit significant differences that correlate with the origin of the IgG. The glycan profiles for rabbit and goat IgGs demonstrate increased complexity, particularly for goat IgG, which displays several additional sialic acid-containing glycans. Consequently, a column capable of providing a high resolution for sialic acid-containing glycans is anticipated to facilitate a more detailed investigation of the glycan profile in particular for goat IgG.
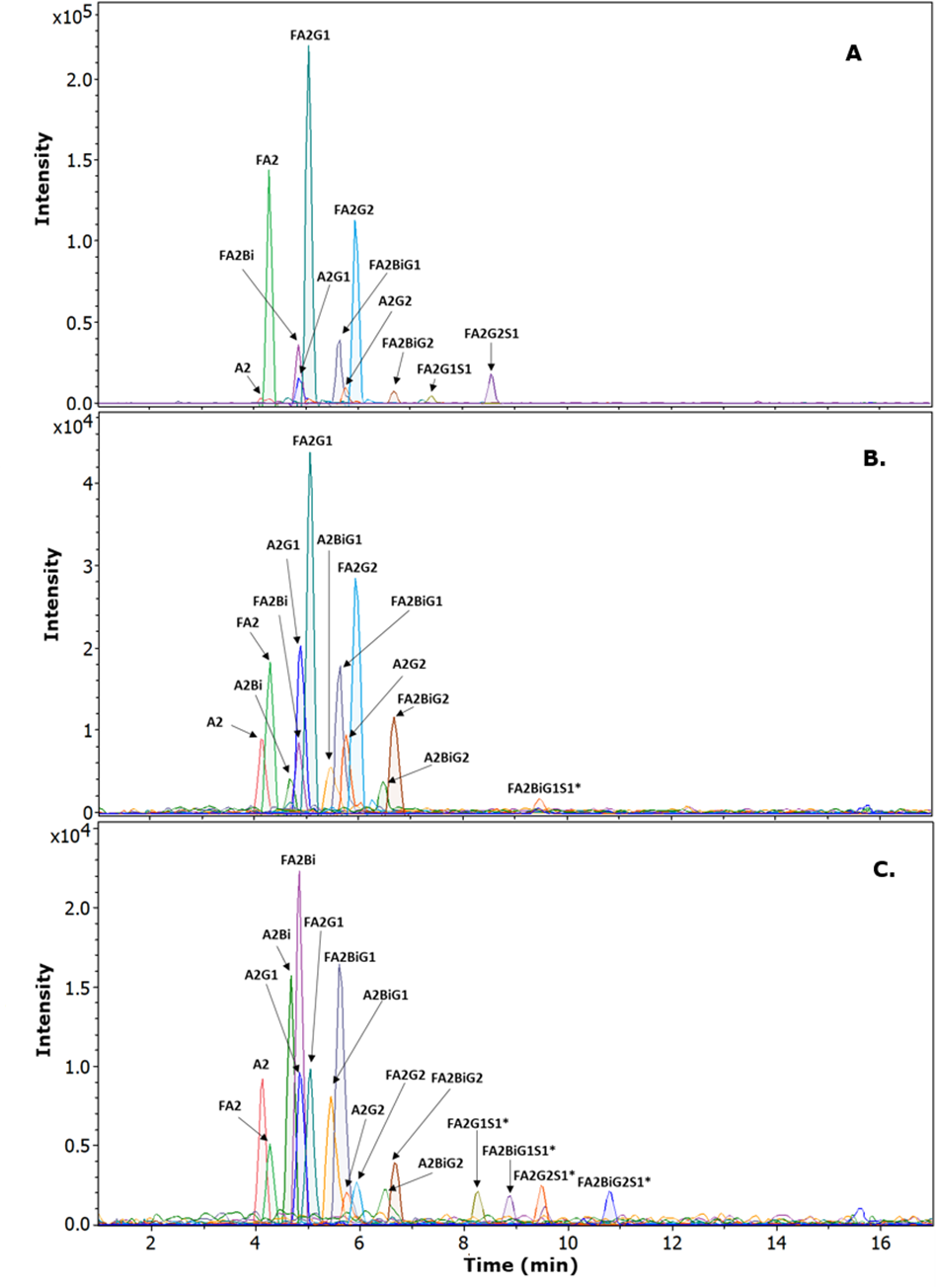
Figure 5.Chromatograms for N-glycans released from IgGs of different origin on Column C (PC on 2.7 µm SPP 160 Å, 150 x 2.1 mm, Ascentis® Express 160 Å ZIC®-cHILIC). Traces represent combined extracted ion chromatograms (EIC) for the main observed glycans and their ion species [M+2H]2+, [M+H+Na]2+ and [M+2Na]2+.
Details of observed masses for [M+2H]2+: A. Human IgG; B. Rabbit IgG; C: Goat IgG.
Conclusion
Three UHPLC columns with zwitterionic (HILIC) functionalization on two silica particle types were investigated for glycan analysis of different IgGs. Informative glycan profiles of the complex mixtures derived from the IgGs were successfully obtained across all tested HILIC columns. The columns featuring phosphorylcholine functionality (PC, ZIC®-cHILIC type) demonstrated higher retention and improved resolution compared to those with sulfobetaine (SB, ZIC®-HILIC) functionality. Therefore, PC columns are recommended as the preferred stationary phase for further optimization and application in glycan profiling based on the findings of this study. Additionally, the PC columns provided superior separation of complex glycans, including both fucosylated and non-fucosylated glycans, as well as sialylated and non-sialylated glycans. The new Ascentis® Express 160 Å ZIC®-cHILIC column is utilizing superficially porous particles (Fused Core®) with a pore size of 160 Å and does exhibit better resolution than the fully porous particles under the tested conditions, while also reducing the retention time window and overall analysis time enabling a faster and more efficient glycan profiling.
Related Products
HPLC Columns
Reference Materials
Solvents and Reagents
References
To continue reading please sign in or create an account.
Don't Have An Account?