Protein Pull-Down Techniques
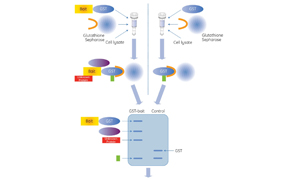
The pull-down assay is designed to determine the interaction of two or more proteins. With affinity pull-down assays, a ″bait″ protein is tagged and captured on an immobilized ligand (support beads) by covalent attachment or through an affinity tag such as immobilized metal affinity chromatography (IMAC). Common bait protein fusion tags include Glutathione S-transferase (GST) and histidine-tagged proteins. The bait protein forms a complex which is then incubated with a protein source such as cell or tissue lysate. The proteins of interest are eluted from the beaded support through a series of washes and collected by centrifugation. The affinity pull-down method of purification and detection is different from immunoprecipitation (IP) and Co-IP assays in that it is not an antibody to antigen interaction. With all pull-down assays, the purified sample is often analyzed by SDS-PAGE, mass spectral analysis, and western blot detection for further downstream analysis.
Featured Categories
Cytiva™ technologies have enabled breakthroughs in science and medicine that have shaped today's biotechnology industry.
Optimize protein purification with our range of affinity resins and buffers for multiple tags...
Streptavidin and avidin resins and prepacked columns for small- to large-scale...
Affinity GST Pull-Down Applications
Glutathione S-transferase (GST) is a 211 amino acid protein that is found in most organisms. GST is often integrated into expression vectors to produce the fusion tag within E. coli protein expression systems. The GST-tag is a large protein tag, approximately 26 kDa, and can be expressed in bacteria, yeast, mammalian, and insect cells. The GST-tag is advantageous when protection of the recombinant protein from intracellular protease cleavage is required, and when there is a need to enhance the solubility of the tagged protein. In addition, the GST protein provides a high-affinity tag for easy purification and for pull-down experiments utilizing inexpensive affinity resins that are performed in mild elution conditions.
Co-Immunoprecipitation
The pull-down assay is ideal for verification of co-immunoprecipitation results and an excellent method of identifying unknown protein-protein interactions along with the activation status of specific proteins. Unlike affinity pull-down methods, IP uses antibodies to bind to and isolate antigens from complex biological samples.
Co-IP refers to the use of an antibody to bind an antigen within a multiprotein complex. The complex is then captured with the addition of protein A or protein G medium. The high affinity of protein A/G to the Fc region of polyclonal and monoclonal IgG-type antibodies makes them a critical component in the purification of the protein or protein complex of interest. The use of protein pull-down methods is an essential tool for investigating protein-protein interaction networks; however, the choice of the specific method will be determined by the researcher’s specific application need.
Visit our document search for data sheets, certificates and technical documentation.
Related Articles
- CsCl gradient centrifugation separates RNA from DNA; differential and density gradient centrifugation techniques explained.
- Centrifugation enables the separation of particles by sedimentation. Learn how to separate particles using a centrifuge and how to use Stokes' law to calculate the velocity of sedimentation.
- Sera-Mag and Sera-Mag SpeedBeads provide cost effective magnetic bead separation technology for molecular biology applications, nucleic acid isolation, and research immunoassays.
- Agarose beads Vs. Magnetic beads in Chromatin Immunoprecipitation (ChIP)
- This page describes immunoprecipitation (immunoaffinity or pull-down techniques).
- See All (10)
Related Protocols
- Techniques for protein antigen molecular weight determination, protein interactions, enzymatic activity, and post-translational modifications.
- Immunoprecipitation Kit (Protein G) Protocol & Troubleshooting
- Immunoprecipitation Kit (Protein A) Protocol & Troubleshooting
- Sera-Mag SpeedBeads provide fast and convenient protein isolation; suitable for manual or automated magnetic affinity binding in various applications.
- Nanodisc technology aids membrane protein solubilization, overcoming associated challenges in diverse protein classes.
- See All (13)
Find More Articles and Protocols
How Can We Help
In case of any questions, please submit a customer support request
or talk to our customer service team:
Email custserv@sial.com
or call +1 (800) 244-1173
Additional Support
- Chromatogram Search
Use the Chromatogram Search to identify unknown compounds in your sample.
- Calculators & Apps
Web Toolbox - science research tools and resources for analytical chemistry, life science, chemical synthesis and materials science.
- Customer Support Request
Customer support including help with orders, products, accounts, and website technical issues.
- FAQ
Explore our Frequently Asked Questions for answers to commonly asked questions about our products and services.